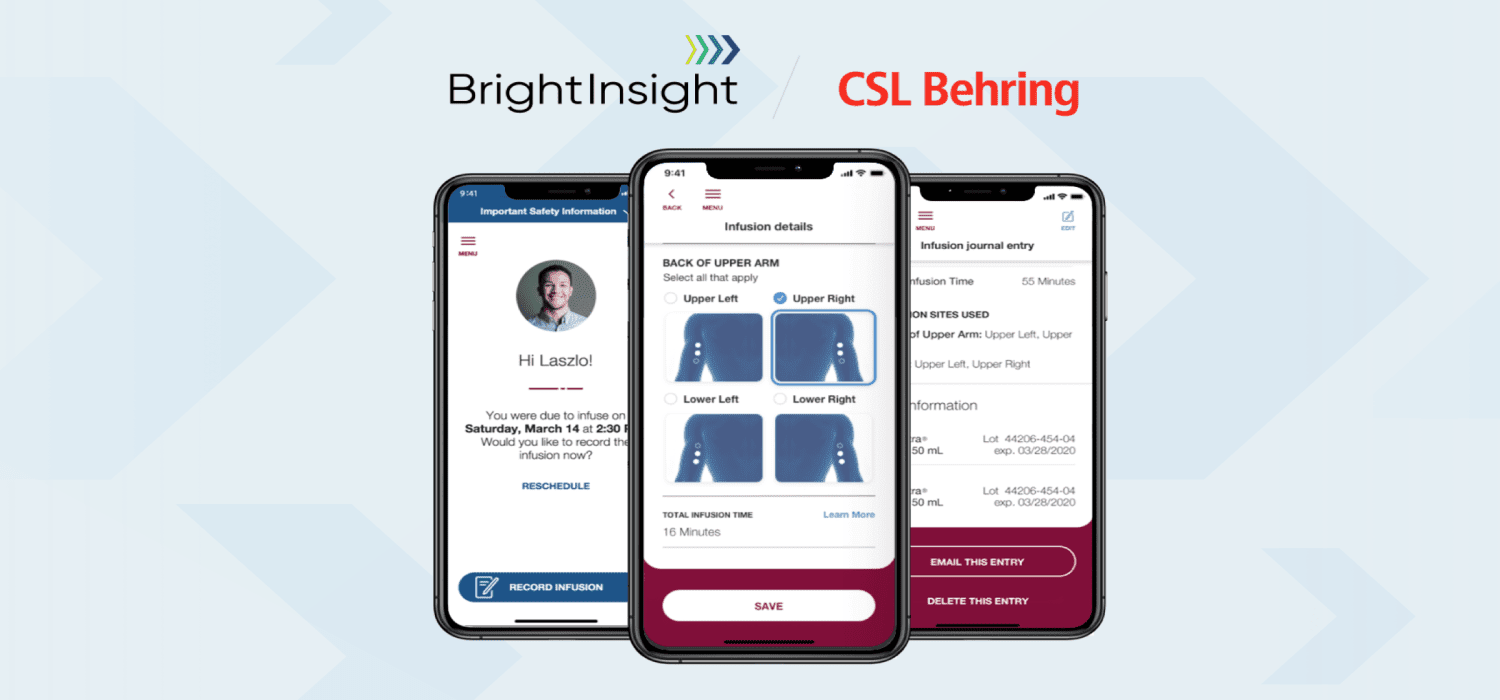
BrightInsight, Inc., provider of the leading global platform for biopharma and medtech regulated digital health solutions, today at HLTH 2022 announced an enterprise agreement with CSL Behring, a leading biotechnology company. Following the swift and successful launch of CSL Behring’s Hizentra® app, BrightInsight will help CSL Behring build, launch, and maintain digital solutions for its late stage and marketed rare disease therapies. The expanded partnership includes plans for additional apps and geographic expansion. BrightInsight will also serve as legal manufacturer of record (LMR).
CSL Behring and BrightInsight began their partnership in May 2020 when CSL Behring selected BrightInsight to develop an app to enhance treatment experiences for adult patients taking Hizentra for two rare diseases, Primary Immune Deficiency (PI) or Chronic Inflammatory Demyelinating Polyneuropathy (CIDP). Launched in less than 6 months, the Hizentra® app has an app store rating of 4.5+ stars and supports patients as they transition from in-clinic to at-home self-administered infusions of Hizentra. The company has captured over 800,000 electronic patient reported outcomes (ePROs) and over 160,000 at-home self-administrations of the drug. The Hizentra app has been well-adopted by the brand’s U.S. patient population and offers features that have resulted in high user retention that is nearly double the industry average.
“CSL Behring is an outstanding partner and truly understands digital’s power to provide insights that can improve outcomes, increase medication adherence and enhance the patient experience,” said Kal Patel, M.D., CEO and co-founder of BrightInsight. “We are thrilled to expand our partnership and introduce new digital health products across CSL Behring’s portfolio of rare disease medicines, leveraging our platform and out-of-the-box solutions to drive speed to market, expand globally and deliver value for patients and brands.”
BrightInsight provides the leading global platform for biopharma and medtech regulated digital health solutions. When speed matters, we help companies accelerate time to market for regulated digital health offerings across therapeutic areas, including apps, healthcare provider interfaces, analytics dashboards, algorithms, medical devices, connected combination products, diagnostics and Software as a Medical Device (SaMD). BrightInsight replaces the need for lengthy and complex ‘build from scratch’ implementations by offering configurable software solutions and a proven platform built on Google Cloud under a Quality Management System to support global security, privacy and regulatory requirements. When building digital health products on the BrightInsight Platform, compliance is future-proofed as intended use changes scale across geographies.
For more information, visit BrightInsight’s website and Blog, and follow us on LinkedIn, Twitter, and Instagram.
