In late 2019, one of the world’s largest biotechnology companies made a decision that, little did it know, would help usher forward a new era of pain management.
Amgen’s choice to mostly exit brain drug research wasn’t out of the blue. Neuroscience, even in the labyrinthine world of drugmaking, is notoriously risky and expensive, and its challenges have convinced many developers their dollars would be better spent elsewhere. After Amgen joined that list, some of its laid off staff found new roles just down the street from the company’s Los Angeles-area headquarters, at a startup formed by venture capitalists at Westlake BioPartners.
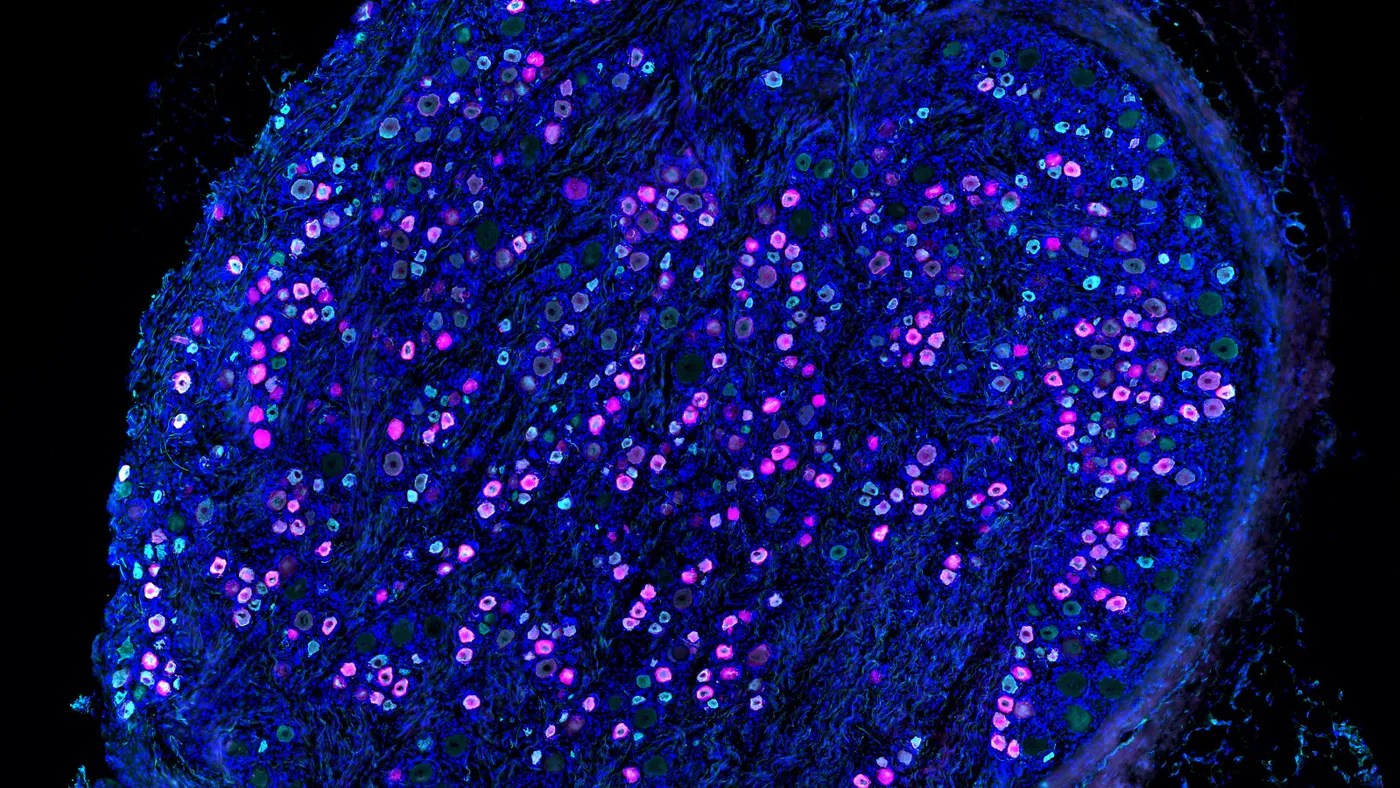
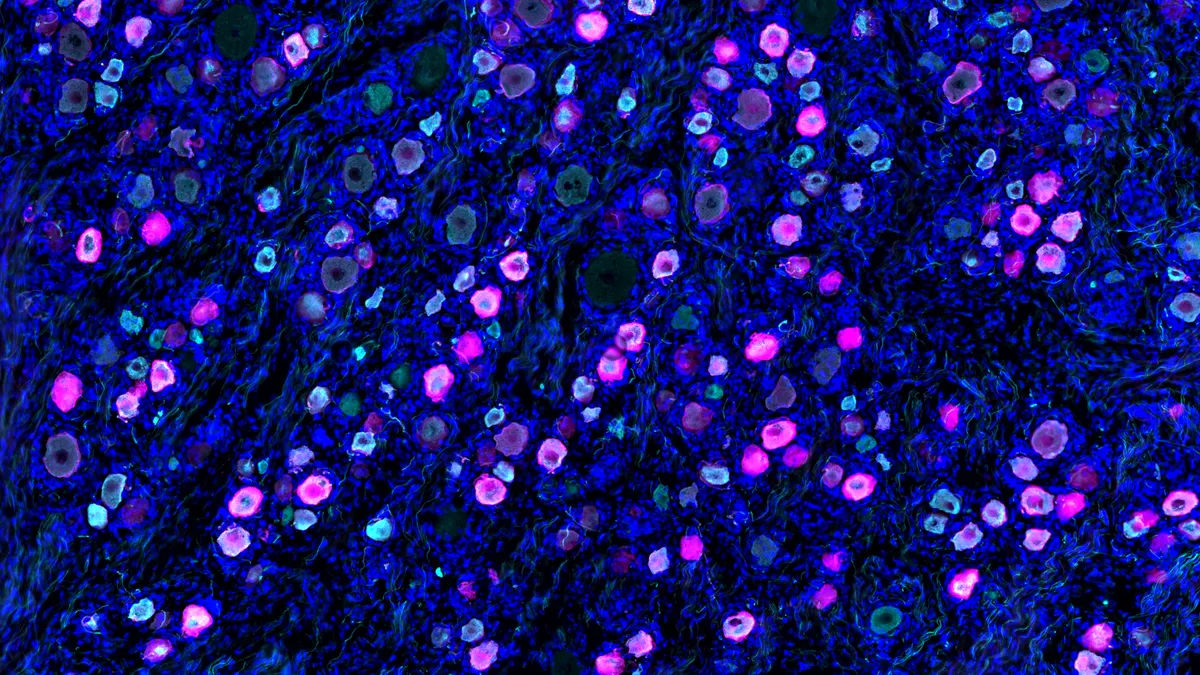
For Westlake, the moment was right to jump into a branch of science three decades in the making, where researchers had uncovered that certain nanoscopic tubes in the outer layers of cells play a key role in pain. These “sodium ion channels” are tricky to study and even harder to control. But years of slow, grueling experimentation was finally paying off.
Pain, being a virtually universal human experience, has long been a white whale opportunity for drug hunters. If anyone could find a strong analgesic that’s safer than opioids, the social and financial payoffs would be massive. In other words, a smart bet for Westlake — which, by itself and with relatively little funding, got that startup, Latigo Biotherapeutics, off the ground in 2020.
“It was a basic benchtop science exercise, knowing that on the other side there could be a GLP-1-like, decabillion-dollar market,” David Allison, a managing director at the firm, said in an interview in late November.
Yet, when the Westlake team pitched Latigo to fellow investors the following year, Allison recalls a less-than-enthusiastic reception. They didn’t care to gamble on pain pills, even novel ones, at a time when cutting-edge genetic medicines were promising to cure an array of devastating diseases. “Most people said: ‘A pain company in 2021? You’re crazy.’”
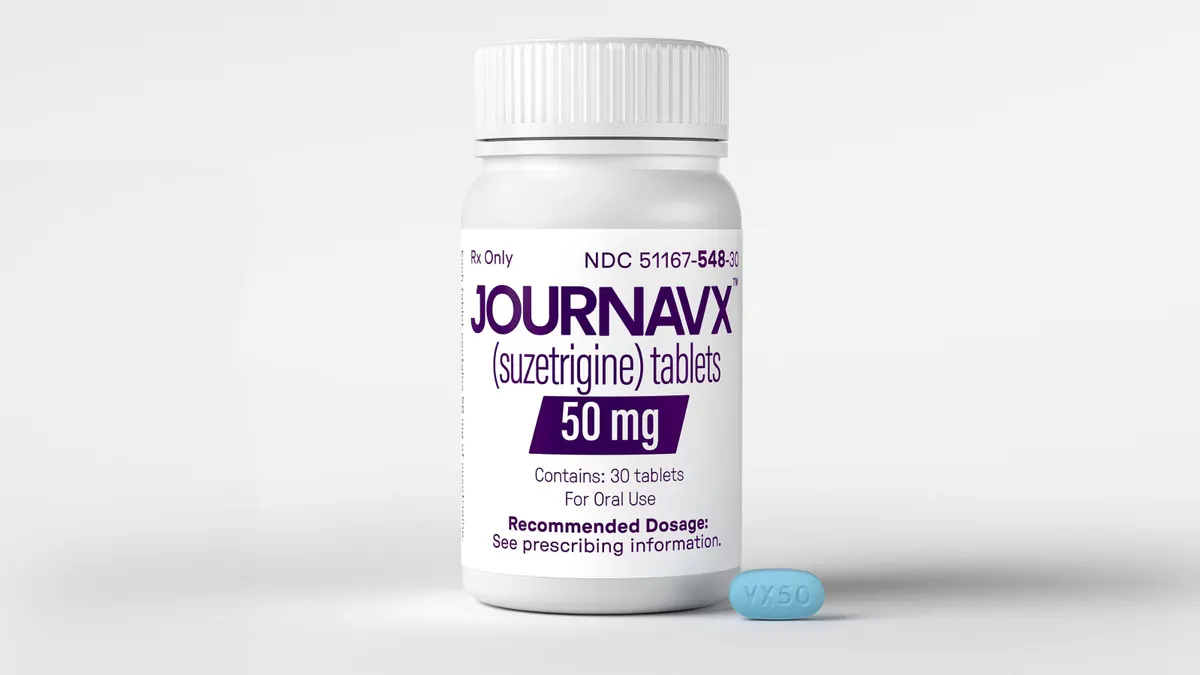
Five years later, Westlake is vindicated. Latigo raised $150 million from a funding round announced last March, right as Journavx — a first-of-its-kind ion channel blocker from Vertex Pharmaceuticals — launched in the U.S. Though it took a quarter century to bear fruit, Vertex’s work has started to revive a graveyard area of drug development. A handful of companies are following in its footsteps with similar medicines, including pharmaceutical giant Eli Lilly.
Pain drug research now faces a pivotal moment. For major investments to continue, the field must soon deliver assets worth rallying around beyond Journavx and its closest rivals. That’s proving difficult, as testing new painkillers remains an all but insurmountable task.

Journavx itself is not yet the roaring success some had hoped. Despite being exquisitely designed, it appears about as potent as a weak opioid, leaving doctors unsure when and where it’s most useful and researchers pondering whether drugs like this have a firm limit to their power. To craft something superior is a daunting enough premise that it could dissuade more developers from even trying.
On Wall Street, the early buzz surrounding Journavx has quieted amid disappointing first-year sales. Analysts don’t foresee revenue drastically ramping until the drug gets OK’d for a wider range of painful conditions.
“Excitement is definitely there,” said Phil Nadeau, an analyst at TD Cowen who covers Vertex. “But, ultimately, we'll need some examples of successful commercial development for that enthusiasm to persist.”
A rare victory
Pain is exceedingly tough to define and defeat, a gnawing reminder of how much we still don’t understand about our own biology. Until Journavx, there hadn’t been a truly innovative medication for it cleared in the U.S. in over two decades.
Journavx won approval in early 2025, as a treatment for the short-lived “acute” pain typically felt after an accident or operation. It was, as Nadeau describes, a “watershed moment,” providing a desperately needed non-addictive option at a time when millions of Americans each year report misusing a prescription opioid. Overdose deaths involving these drugs are remain in the thousands annually.
Doctors are now using Journavx after everything from knee replacements to shoulder surgeries to wisdom teeth extractions. In April, total prescriptions surpassed 1 million.
“We've seen great receptivity, particularly in those institutions keen to move away from opioids as much as possible,” said Duncan McKechnie, who, as Vertex’s chief commercial officer, is overseeing the rollout. “We're really pleased with the progress so far, but we're a long way to go to transform the treatment of pain worldwide.”
High demand hasn’t made the launch frictionless — or cheap. Last year, Journavx was the main culprit behind a 20%, or almost $300 million, surge in Vertex’s overhead costs. For the first time, the company turned to marketing tactics from big pharma’s playbook. It bought ads for TV, Instagram and YouTube, and partnered with high-profile athletes like Jayson Tatum and Alex Smith, and is in talks with Lindsey Vonn.
Spending looks poised to grow further, as Vertex just a few weeks ago doubled the size of its Journavx sales force, from 150 to 300, in an effort to sway more physicians to routinely reach for the pill.
While many drug launches come with a learning curve, Vertex doesn’t seem to be driving sales fast enough to appease shareholders. Journavx generated just shy of $90 million in its first year on the market. Over most of that period, its performance fell below analyst forecasts.

“Certainly, people don't feel as bullish about the drug as they did a year ago,” said Paul Matteis, an analyst at Stifel.
To Allison, Journavx has done as well as anyone could have anticipated. Even so, an argument could be made that if the drug were under the banner of a company with a sweeping, more primary care-leaning sales team, then it would be closer to meeting the blue sky, “zero-to-a-billion-in-how-many-quarters” bar set by some.
“Investors are looking at this and saying, ‘If this was in the hands of Lilly or J&J or Pfizer, it would be like selling Viagra. It would fly off the shelves,’” Allison said. “I think there's just a little bit of a mismatch in expectations.”
Vertex will start keeping more of each Journavx sale next year, as it pulls back on the free drug giveaways used to build demand ahead of insurance coverage.
Analysts, though, see that as a drop in the bucket compared to the money that would flood in from an approval for chronic pain, which affects far more people — around one in four Americans — and where the long-term use of opioids is of great concern.
Because acute pain prescriptions are, by nature, very short, drugs in this setting must be quickly reimbursed and doled out on an enormous scale to drive meaningful revenue. “All that flips with chronic,” Matteis said. So, “everyone still believes that’s a huge market, myself included.”
The odds of Journavx entering this lucrative market narrowed after it failed a sciatica study last summer. Its future as a chronic pain drug now rests on a late-stage clinical trial that’s focused on the nerve pain caused by diabetes and set to produce data in 2027.
“Vertex is in a tough spot,” Nadeau said.
With $13 billion in cash and cash equivalents on hand, plus an arsenal of cystic fibrosis medicines that print $3 billion a quarter, Vertex has deep enough pockets to trudge through such a hazardous market. The same likely wouldn’t be true for smaller biotechs.

A one-two (or three) punch
Journavx has another, paradoxical problem: it’s remarkably safe.
This should be a clear advantage, except that almost nothing is so straightforward in the nervous system. Indeed, some of the best neuron-stabilizing medicines are also the “dirtiest,” meaning they’ll brazenly shack up with any number of molecular partners.
Journavx, by contrast, is calibrated to hit just one target, a sodium channel called NaV1.8, with missile-like precision. It’s the biological equivalent of using a scalpel instead of a bone saw. But while one is more elegant, each carries tradeoffs. “The cleanness might be its Achilles heel, which is that it probably doesn't have the same oomph as an actual opioid,” Matteis said.
Whether any more juice can be squeezed out of NaV1.8 inhibitors has been a nagging question for Stephen Waxman, a neurology professor at Yale University whose decades studying ion channels and a rare disease known as “Man on Fire syndrome” has made him a preeminent voice in pain science. Scouring for answers, he and his colleagues devised a system to effectively speak with pain-sensing cells.
Using an ultra-thin glass pipette equipped with an electrode, they connected rat neurons to specialized computers capable of replicating the electrical pulses emitted by ion channels. In real time, the computers could tune pulses up and down to determine how much NaV1.8 blocking is necessary to stifle pain signals.
They found reducing the current by as little as 25-50% was enough. Put more plainly: a drug doesn’t have to perfectly neutralize the channel to offer adequate pain relief.
That wasn’t all. A separate Harvard Medical School-led study observed that a small fraction of neurons continued firing pain signals even when most, if not all, of their NaV1.8 activity had been muted. The results add fuel to a growing suspicion that, at least in some cells, targeting NaV1.8 alone only goes so far. Vertex’s own studies hinted as much. A drug seen as a Journavx successor failed in a crucial study last summer. To some analysts, that outcome not only diminished the outlook for the company’s pain business, but confirmed developing a better molecule will be much harder than previously thought.
While such a challenge could scare away competitors, it also presents an opening. Researchers suspect a cocktail of ion channel manipulators may be able to break through the NaV1.8 ceiling. They’re now exploring layered approaches, regulating not only that protein but other immediate members of its family, like “1.7” and “1.3,” as well as more distant relatives like potassium channels.
“We're not looking for a dirty drug so much as, how can I put it, a controlled mess,” Waxman said.
“There are a large number of combinatorial possibilities,” he added, “so the work goes slowly. But that's one way we're moving ahead.”
Ken Harrison sees drug combinations as the next logical step in pain investing. As a senior partner at Novo Holdings, the controlling stakeholder of Ozempic-maker Novo Nordisk, Harrison is part of a firm that backed at least four startups in the pain space, including SiteOne Therapeutics, which Lilly purchased in 2025 through a deal worth up to $1 billion.
Vertex and SiteOne previously agreed to collaborate on non-opioid painkillers directed at NaV1.7. In an earnings report last week, Vertex made note that it continues to advance preclinical NaV1.7 inhibitors, to use alone or pair with NaV1.8 drugs, for the treatment of acute and neuropathic pain.
At least half a dozen more companies have NaV1.7 research programs. One, Xenon Pharmaceuticals, is an ion channel specialist whose stock surged almost 60% over the past 12 months thanks to positive data for a closely watched epilepsy and depression treatment.
In pain, Xenon has pushed a NaV1.7 clogger along with a potassium channel opener into early human studies. The Canadian drugmaker touts “probably the most underrated pipeline program in pain right now,” Matteis said.

An investable space
The way Harrison tells it, SiteOne came on Novo Holdings’ radar not long after forming in 2010. The firm kept a watchful eye from afar, for 14 years, before jumping in to lead the biotech’s $100 million funding round.
Novo Holdings made the leap partly because of the precedent Vertex was setting, partly because of SiteOne’s leadership and encouraging early data. There was a pull, too, in the stories Harrison’s team heard as they conducted due diligence. One stirring conversation was with an ER doctor who guessed that, across 20 years of treating patients, he had written north of 10,000 opioid prescriptions.
Statistically, some of those patients became addicted. Some probably died. “That haunted him,” Harrison said. “And I don't think he's alone with that vignette. Many physicians are interested in other agents.”
A patient population in the tens of millions, who urgently need new, safe medicines, would usually be an irresistible draw for drugmakers. Pain, however, isn’t like most areas of development, where animal tests or molecular markers can offer at least some reliable clues that a medicine might work in people. It also varies widely not only from species to species, but person to person, a black box that has thwarted an immeasurable number of potential treatments.
This underlying peril is reflected in deals and dollars. Outside of a novel class of migraine therapies that emerged in the 2010s, big pharma hasn’t spent much on acquisitions in this space in a while. The nine-figure financings that propped up Latigo and SiteOne are also anomalies. Venture capital funding for other biotechs chasing pain has been paltry.
“I've had a decision maker in biopharma tell me, “You'd have to be crazy to invest in working on pain,’” Waxman said.
They’ve got examples to draw from. Though a different disease, schizophrenia also affects a wide swathe of the population and remains an ever-tantalizing opportunity for drugmakers. Yet a highly anticipated medicine Bristol Myers Squibb paid billions to acquire hasn’t broken through in a market flush with generics, frustrating investors and leading to doubts about its commercial potential.
Harrison said late last year that while his team remained intrigued by pain opportunities, they’re unlikely to bet again on a NaV1.8-centric company. Pain drug combinations are more attractive, he added, though “I can't say we’ve found one we're interested in investing in quite yet.”
More broadly, the headcount of developers , and recent entry of a large, resource-rich player in Lilly, also makes this a more complicated investment prospect than just 18 months ago. Starting a company like Latigo would be tougher, according to Allison, as it needs to be “really differentiated, almost like the obesity space.”
Still, biotech investments come in momentous waves. A single breakthrough — be it from Vertex, Lilly or a small startup — could yield the next surge.
“Sometimes venture investors make sheep look like independent thinkers,” Allison said.
Correction: A previous version of this story misstated the number of Journavx prescriptions written so far and misattributed NaV1.8 research done by Yale University.