Dive Brief:
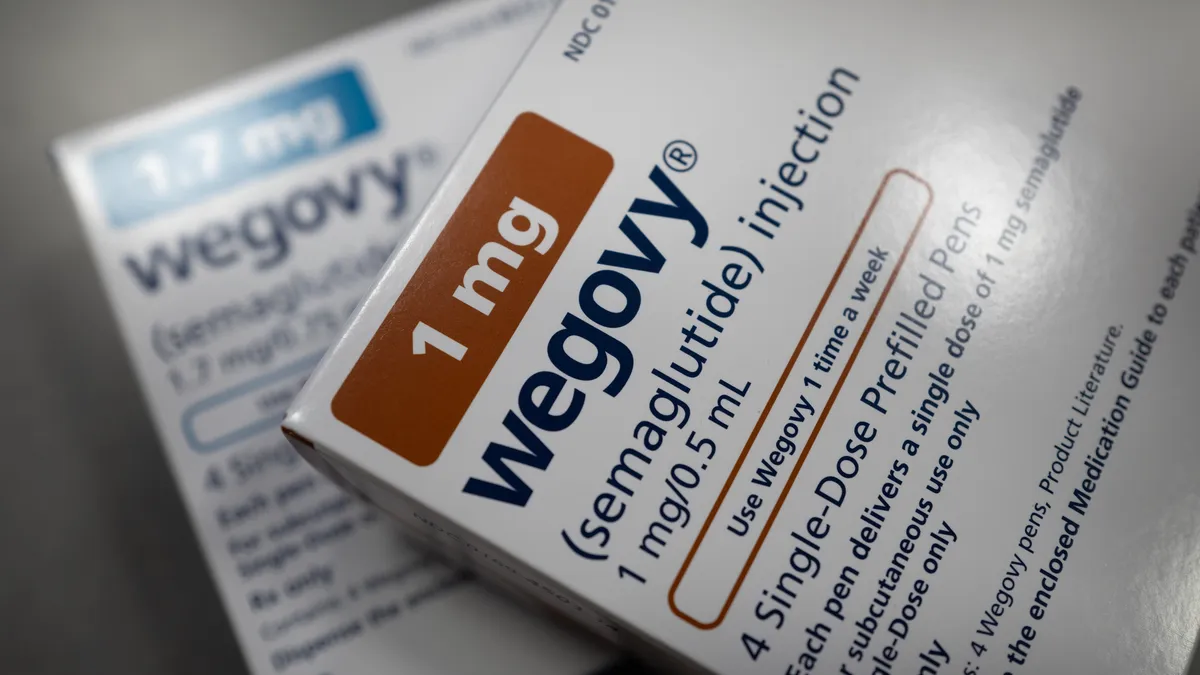
- Veru’s muscle-preserving obesity drug enobosarm, when combined with Novo Nordisk’s Wegovy, may have less gastrointestinal side effects at the dose planned for pivotal testing than Wegovy alone, according to safety results from a Phase 2 clinical trial the company released Wednesday.
- Fewer people receiving either of two doses of enobosarm and Wegovy experienced nausea or heartburn than those taking just Wegovy, and, at the enobosarm dose planned for Phase 3 testing, fewer had diarrhea or vomiting, Veru said. However, the company reported more volunteers given enobosarm had elevated liver enzymes, although levels subsequently dropped during the trial.
- Veru shares rose significantly in morning trading Wednesday, although at around 60 cents a share they remain well below the $1.28 hit before release of topline weight-loss data in January. That data suggested enobosarm helps with body composition, but doesn’t increase weight loss when used with Wegovy.
Dive Insight:
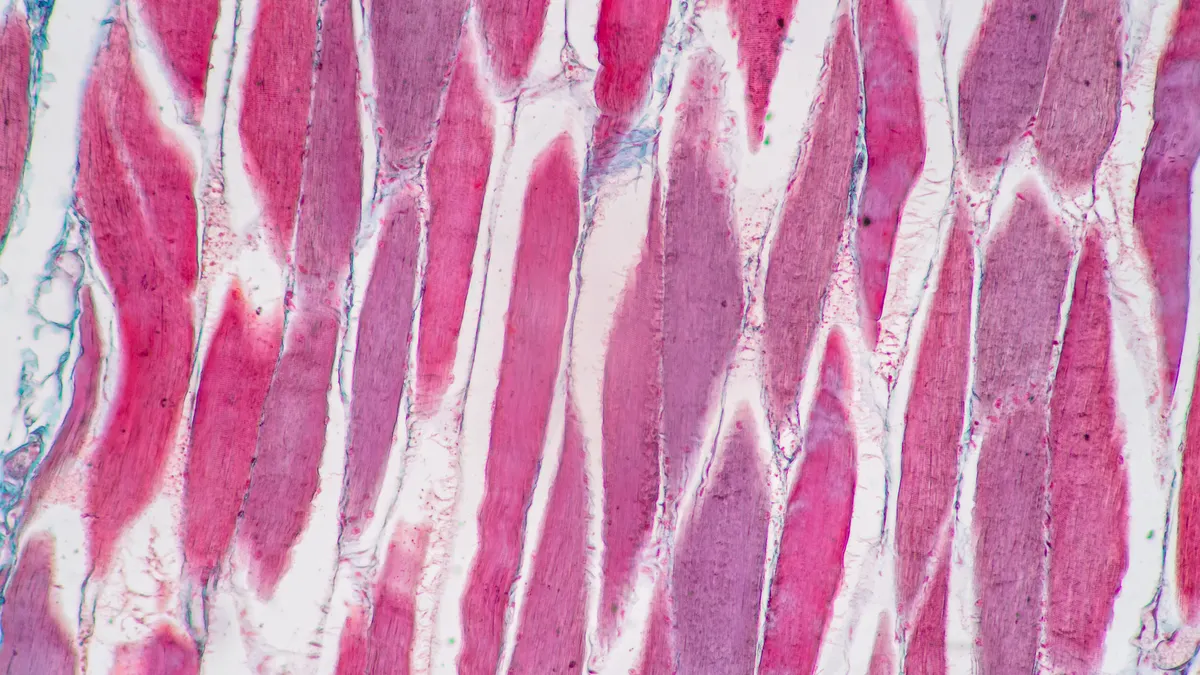
The safety data from the trial had remained “blinded” to the company and researchers until participants could complete an extension to the main 16-week stage of the trial. The 168-person trial sought to show that adding enobosarm to Wegovy could result in less loss of lean muscle mass than Wegovy.
Body composition is an emerging issue in obesity treatment as Wegovy and Eli Lilly’s Zepbound also cut lean muscle mass alongside fat, which in older patients could raise the risk of premature frailty. Veru’s trial focused specifically on people with obesity at least 60 years old.
While adding enobosarm to Wegovy didn’t increase weight loss versus Wegovy and a placebo, it kept lean body mass loss to 1.2%, significantly less than the 4.1% lean body mass loss seen with the Wegovy plus placebo arm. Fewer people who took enobosarm and a placebo also saw substantial losses in their stair-climb power, a measure of physical function.
Veru tested two doses of enobosarm, 3 and 6 milligrams daily, alongside weekly shots of Wegovy. Based on the data seen so far, Veru will take the 3 milligram dose into a Phase 3 trial because the 6 milligram dose “did not provide any additional benefit.” At that 3 milligram level, only one trial participant experienced elevated liver enzymes — a mild case in which the individual’s levels returned to baseline while they were still taking the drug.
Meanwhile, the company also said it is preparing a “novel, patentable, modified release oral formulation” for Phase 3 testing and potential commercialization that will undergo Phase 1 trials in coming weeks. The company said it believes the formulation patent from the new version will extend its market exclusivity to 2045.
Enobosarm has been tested in the past as a drug to prevent muscle wasting in people with lung cancer and other disease settings.