Dive Brief:
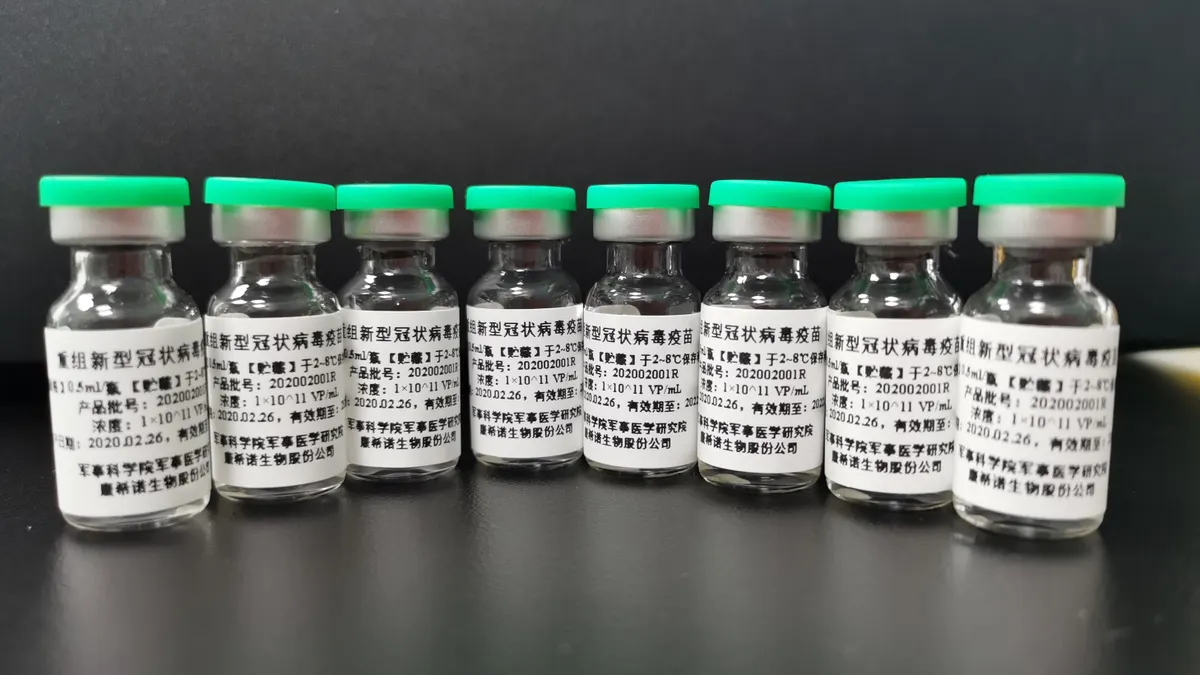
- CanSino Biologics, one of the first biotech companies to test a coronavirus vaccine in humans, has turned its research progress into a lucrative market debut on the Shanghai stock exchange, raising roughly $750 million in a stock offering Thursday.
- Shares in the Tianjin, China-based drugmaker rose nearly 90% in the stock's first day of trading on the Nasdaq-styled exchange, also known as the STAR market. CanSino shares are also listed on the Hong Kong stock market.
- CanSino's coronavirus vaccine has advanced through Phase 1 and 2 studies, results from which have shown the shot to be generally safe at lower doses and capable of triggering an immune response. But the company has had difficulty finding a country in which to launch a large-scale efficacy trial, as cases have waned in China. Saudi Arabia reportedly agreed last week to let CanSino run a 5,000-participant trial there.
Dive Insight:
Lesser known in the U.S. and Europe, CanSino is one of the most advanced Chinese vaccine developers, and has capitalized on that status to secure $750 million more for its research.
The company said the funds would be put toward constructing a manufacturing plant, as well as vaccine testing and development of a "cold chain logistics system."
CanSino currently has a production plant in Tianjin that it says is capable of making 70 million to 80 million vaccine doses a year. But more capacity will likely be needed if further testing proves the company's shot is protective. In June, the Chinese military approved the vaccine for use in soldiers based on results from the first two studies CanSino ran.
The trials showed the vaccine did induce an immune response, although the resulting antibody levels appear to be weaker that those generated by candidates advanced by other companies like Moderna, Pfizer, Novavax and AstraZeneca.
A sizable portion of people who received CanSino's vaccine also had preexisting antibodies to the non-infectious cold virus the biotech uses to shuttle into cells the genetic instructions for a crucial coronavirus protein. Higher levels of those antibodies weakened the ability of the vaccine to trigger immune defenses against SARS-CoV-2.
Preexisting immunity to the cold virus in question is thought to vary by region, but widespread exposure among a population could potentially make CanSino's product less attractive.
Phase 3 trials would provide more information, but CanSino has only just begun to start those studies. The planned trial in Saudi Arabia is reported to target only 5,000 participants, much smaller than the 30,000-volunteer tests other companies are currently planning in the U.S. and Europe.
CanSino has also received approval to test its vaccine in Canada, although problems exporting the required doses seem to have delayed its initiation.
Other Chinese companies have looked to outside countries to complete their vaccine testing. Sinovac, for instance, has secured approval to test its shot in Brazil.