Relay Therapeutics, a six-year-old biotech trying to make better drugs by studying how the proteins they target move, on Friday released the first clinical trial results for its most advanced treatment.
On one score at least, Relay seems to have had some success. The company's drug, a cancer medicine known as RLY-4008, appears less toxic than its would-be competitors. But RLY-4008 still caused other side effects, particularly at a more frequent dosing schedule that's been discontinued. And the preliminary data disclosed by Relay, while promising, are too early to prove whether the drug is more effective.
The results are from four dozen patients in a Phase 1 trial and some uncertainty is to be expected as Relay figures out the best dose of its drug. But the data are nonetheless a test case of the company's pitch that its technology can give it a leg up over other drug developers.
Founded in 2015, Relay aims to pair established drug-hunting techniques with simulations of how proteins move and shape-shift — valuable information for designing medicines. The company's methods borrow from, and extend the structure-based approach Vertex Pharmaceuticals first took to drug design and popularized decades ago. (Mark Murcko, a veteran Vertex researcher and executive, was one of Relay's founders.)
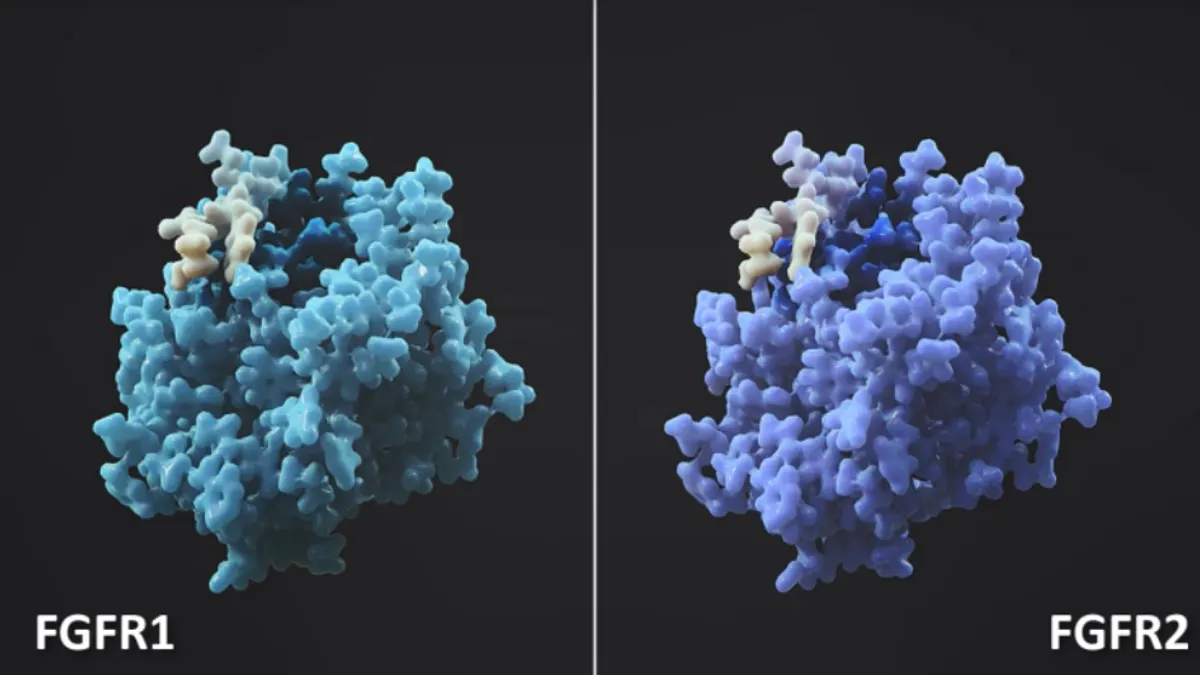
In the case of RLY-4008, Relay used protein motion models to identify key differences in the movements of two related proteins called FGFR1 and FGFR2, the latter of which is frequently mutated in certain cancers. Though the two are seemingly identical to each other when frozen in crystal, FGFR1 has a segment that extends outward from the protein more often than FGFR2.
Relay scientists used that discovery to design RLY-4008 to target only FGFR2 and, in theory, avoid FGFR1 and other related proteins.
Three FGFR inhibitors are already approved: Johnson & Johnson's Balversa in bladder cancer as well as Incyte's Pemazyre and QED Therapeutics' Truseltiq in a type of bile duct cancer called cholangiocarcinoma.
But all three can inhibit FGFR1, which is needed by the kidneys to reabsorb phosphate from the blood. By blocking FGFR1, the drugs commonly cause high phosphate levels, which in testing led patients to pause or stop treatment.
Relay's results were different. In the Phase 1 study, which mostly included patients with cholangiocarcinoma, the rate of hyperphosphatemia was much lower than observed in clinical trials of Pemazyre, Trulsetiq and an experimental FGFR inhibitor called futibatinib.
"Despite hammering FGFR2, we're seeing a rate of hyperphosphatemia that's quite low and clinically irrelevant," said Don Bergstrom, head of R&D at Relay, in an interview. "It's transient and doesn't require medical management."
Treatment with RLY-4008 also led to much lower rates of diarrhea, another common side effect with FGFR inhibitors.
Yet while rates of both side effects were lower, they still occurred with RLY-4008, particularly when the drug was taken twice a day. Relay has stopped further testing of that dosing in favor of once daily, but the data could suggest RLY-4008 is still inhibiting other FGFR proteins, just much less readily.
"While at some of the higher doses we may have been starting to maybe tickle FGFR1 a little bit, I think what this is telling us is that really, especially in the once-daily dosing schedule, we're seeing minimal engagement of FGFR1," explained Bergstrom.
RLY-4008 also caused a number of other notable side effects, including mouth sores, nail toxicities and a skin condition associated with some cancer treatments called hand-foot syndrome. All were reported in testing of the other FGFR inhibitors.
Relay's goal isn't just to design a safer version of available drugs, though. The company aims to prove RLY-4008 is more effective as well.
On that measure, Relay had some promising data to share, but overall results were limited by small sample sizes and follow-up that's still continuing.
Among six patients with cholangiocarcinoma who hadn't previously received FGFR-targeted drugs, treatment with RLY-4008 shrank tumors in three, including one whose tumor was then surgically removed.(Another of the responses, however, was from the twice-daily regimen that Relay has discontinued.)
RLY-4008 was also able to shrink tumors in patients who were previously treated with an FGFR blocker and had developed resistance to treatment, although none were judged to have a confirmed response yet.
Outside of bile duct cancer, Relay observed a partial response in a trial participant with breast cancer who had a different type of FGFR mutation.
"I think this now our beachhead for moving beyond cholangiocarcinoma and moving more broadly into patients with FGFR2-altered tumors," said Bergstrom.
Relay expects to choose a dose for Phase 2 testing by the end of the year, and will disclose updated results early in 2022.
Shares in Relay, which is currently worth nearly $3 billion, remained halted on Friday, although the stock fell by more than 10% on Thursday before the data were released. Results were presented at the virtual AACR-NCI-EORTC Molecular Targets and Cancer Therapeutics conference.