FDA: Page 14
-
Research group says FDA found no misconduct in Pfizer Lyme vaccine trial it helped run
Earlier this year Pfizer removed thousands of participants from a study of its Lyme disease shot over concerns the group, Care Access, wasn't meeting clinical practice standards.
By Delilah Alvarado • Oct. 12, 2023 -
PepGen cleared by FDA to begin study of muscular dystrophy drug
The company expects to deliver first results next year from the early-stage study of people with myotonic dystrophy type 1.
By Kristin Jensen • Oct. 12, 2023 -
FDA denies expanded approval for Alnylam RNA drug
The agency rejected Alnylam’s application for approval of its medicine patisiran in people with a rare heart condition, setting back the company’s plans.
By Jonathan Gardner • Updated Oct. 9, 2023 -
FDA panel finds KRAS drug data unreliable, in blow to Amgen
The negative panel vote likely means Amgen will need more data to support a full approval for its conditionally cleared lung cancer drug Lumakras.
By Ned Pagliarulo • Updated Oct. 6, 2023 -
5 FDA decisions to watch in the fourth quarter
The regulator’s decision on Alnylam’s bid to expand Onpattro’s approval didn’t go the company’s way, but other verdicts await on drugs from Vertex, Bristol Myers, Amgen and Pfizer.
By Ned Pagliarulo , Jonathan Gardner • Oct. 2, 2023 -
ALS drug development
BrainStorm’s ALS therapy not effective, FDA panel finds
Expert advisers to the agency voted 17-1 that Brainstorm's clinical trial data did not show the company's stem cell treatment to be effective for treating ALS.
By Ned Pagliarulo • Updated Sept. 28, 2023 -
FDA staff hold ‘major concerns’ with Brainstorm ALS therapy, documents show
A panel of expert FDA advisers is meeting today to discuss the treatment, and whether Brainstorm’s data provide “substantial evidence” of its effectiveness.
By Ned Pagliarulo • Sept. 25, 2023 -
Intarcia’s diabetes drug-device combo voted down again by FDA panel
The 19-member advisory committee unanimously voted against Intarcia in the review, citing cardiovascular and kidney safety concerns.
By Gwendolyn Wu • Sept. 21, 2023 -
Alvotech gets new FDA review for Humira biosimilar
The regulator has twice rejected Alvotech’s copycat drug due to manufacturing issues with a plant in Europe.
By Jonathan Gardner • Sept. 20, 2023 -
Orchard nears FDA decision on rare disease gene therapy
Three years after gaining European approval, Libmeldy is now under U.S. review with a deadline set for March.
By Jonathan Gardner • Sept. 18, 2023 -
FDA approves updated COVID boosters from Pfizer, Moderna
Pfizer and Moderna, which have seen slowing demand for their coronavirus vaccines, expect their reformulated shots will soon be available in the U.S.
By Delilah Alvarado • Sept. 11, 2023 -
FDA documents hint at ‘uphill battle’ for broad approval of Sage’s depression drug
The newly posted documents show the agency had concerns about the side effects experienced by people with clinical depression who took Sage’s drug, Zurzuvae.
By Jacob Bell • Sept. 1, 2023 -
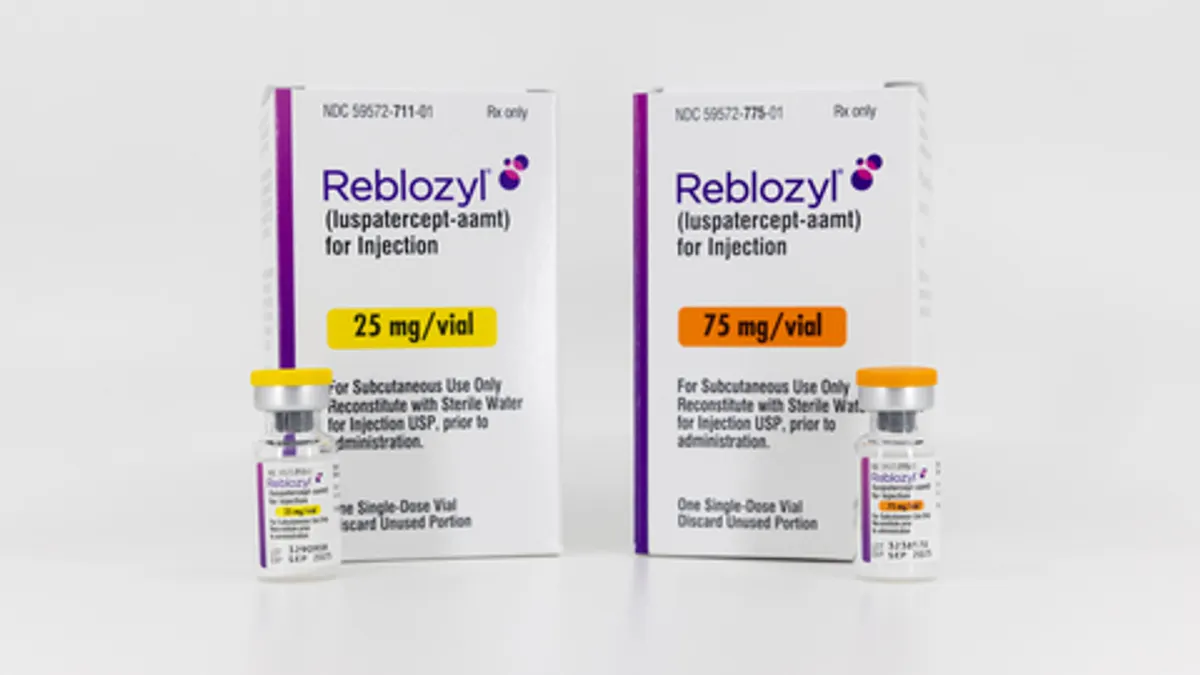
Bristol Myers gets key FDA approval for bone marrow disease drug
A broadened clearance for Reblozyl in myelodysplastic syndromes should help Bristol Myers offset the looming loss of revenue from top-selling medicines set to soon lose market exclusivity.
By Ben Fidler • Aug. 29, 2023 -
FDA approves Novartis’ copycat of blockbuster Biogen drug
According to one analyst, the approval was the last hurdle keeping Sandoz’s Tyruko from directly competing in the U.S. market against Biogen’s inflammation-regulating medicine Tysabri.
By Jacob Bell • Updated Aug. 25, 2023 -
FDA approves Pfizer’s RSV vaccine for use in pregnancy
The shot, called Abrysvo, is the first maternal immunization approved to protect newborns from the respiratory virus in their first months of life.
By Delilah Alvarado • Aug. 21, 2023 -
FDA partially halts leukemia studies of Gilead cancer drug
The hold is the latest setback for a drug that was the center of Gilead’s $5 billion acquisition of biotech Forty Seven in 2020.
By Jonathan Gardner • Aug. 21, 2023 -
Regeneron rebounds to win FDA OK for longer-lasting vision loss drug
The agency cleared high-dose Eylea less than two months after rejecting it, and approved a separate Regeneron drug for an ultra-rare disease as well.
By Ben Fidler • Aug. 21, 2023 -
FDA clears Ipsen bone drug despite questions about its benefits
The mixed results supporting the treatment, Sohonos, have proven a tough case for regulators and led to different outcomes in the U.S. and Europe.
By Kristin Jensen • Aug. 17, 2023 -
Appeals court rules to limit abortion pill access
Federal judges said recent changes by the FDA to loosen prescribing rules for mifepristone should be rolled back. Biotech leaders have warned the case could undermine the agency’s authority.
By Delilah Alvarado • Updated Aug. 17, 2023 -
FDA lifts hold on Arcellx’s Gilead-partnered cancer cell therapy
The agency had paused testing after a patient death, but is now permitting more types of bridging treatment to help keep participants’ disease at bay.
By Jonathan Gardner • Aug. 15, 2023 -
FDA approves bispecific drug from Pfizer for multiple myeloma
Elrexfio is the third bispecific antibody cleared to treat the cancer, and will compete with other therapies that target the “BCMA” protein on myeloma cells.
By Jonathan Gardner • Aug. 14, 2023 -
FDA delays decision on Valneva’s chikungunya vaccine
The agency needs more time to settle with Valneva the design of a post-marketing study for the shot, which could become the first preventive therapy for chikungunya available in the U.S.
By Delilah Alvarado • Aug. 14, 2023 -
J&J joins PARP rivals with narrow FDA clearance in prostate cancer
The agency approved Akeega only for patients with BRCA mutations — another limited OK for a class of drugs that have come under regulatory scrutiny.
By Ben Fidler • Aug. 14, 2023 -
Iveric drug approved for type of vision loss as rival’s safety draws scrutiny
The FDA's clearance of Izervay for geographic atrophy comes as rare side effects have overshadowed the fast launch of a competing treatment from Apellis.
By Ben Fidler • Aug. 5, 2023 -
GSK broadens use of cancer immunotherapy with latest FDA OK
The pharma’s drug Jemperli was cleared for patients with newly diagnosed endometrial tumors, but could soon face competition from Merck’s Keytruda.
By Jonathan Gardner • Aug. 1, 2023