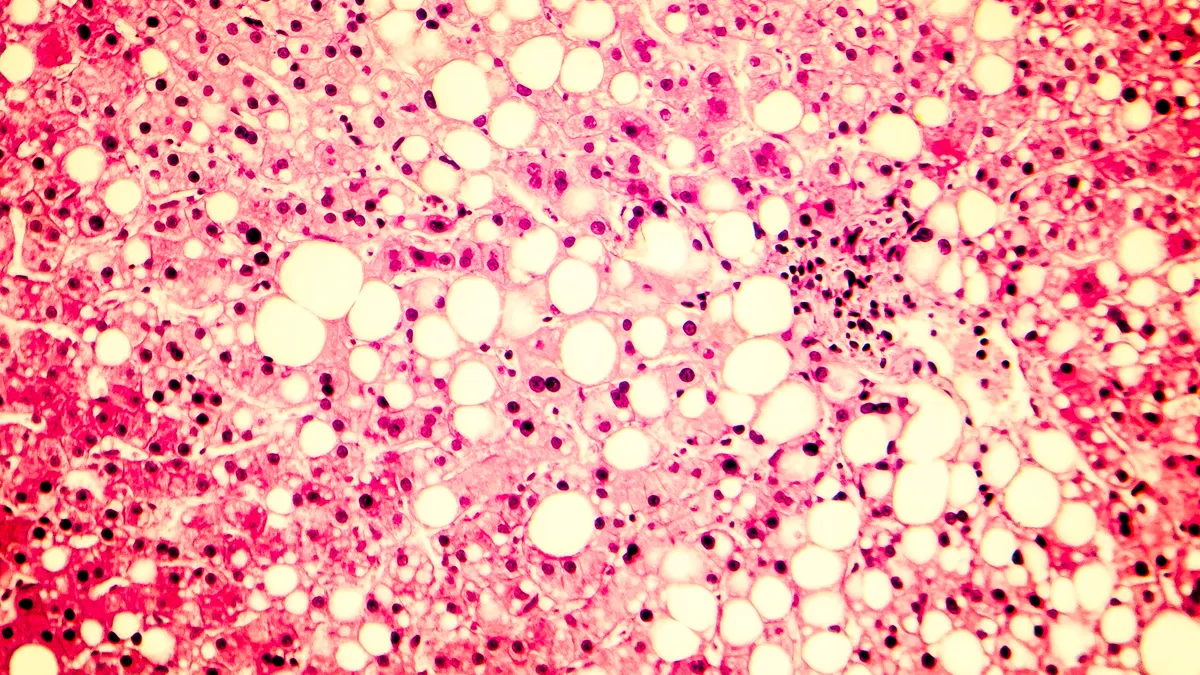
Food and Drug Administration scientists reviewing what could become the first medicine for the fatty liver disease NASH are skeptical the treatment’s safety risks outweigh its “modest” benefits, according to documents made public Wednesday.
The documents were posted online ahead of a Friday advisory committee meeting on the drug, known as obeticholic acid, or OCA, and developed by biotechnology company Intercept Pharmaceuticals. Panelists are being asked whether the FDA should grant the treatment an “accelerated” approval based on biological markers, or defer a decision until Intercept has data from an ongoing Phase 3 trial. That trial is testing the drug’s impact on health outcomes, but results aren’t expected until 2025.
The committee will also discuss whether close monitoring could reduce the risk of drug-induced liver damage — a key concern of regulators — and whether the benefits are worth the risk for a large group of patients with NASH, or non-alcoholic steatohepatitis.
The FDA will decide whether to approve OCA by June 22. It typically follows the recommendations of its advisory panels.
The documents show staff scientists are skeptical OCA is helpful enough to justify a variety of health risks that emerged in testing, from liver and kidney injury to cardiovascular issues like diabetes and dyslipidemia.
For example, FDA reviewers found OCA “exacerbates co-morbidities or creates new ones” for patients already at risk for “metabolic syndrome and its manifestations.” A post-approval plan to lower those risks could be difficult to carry out, and may not eliminate the chance of liver injury. Patients with NASH will require lifelong drug therapy, and compliance to frequent monitoring could be an issue, the reviewers wrote.
While staff scientists acknowledged NASH is an “unmet need,” they also described OCA’s effects as “modest” compared to a placebo. In Phase 3 testing, 21% of patients on a high dose of OCA experienced no worsening of their disease and at least some improvement in liver scarring after 18 months, versus 12% of those in the placebo group. That equated to a difference in risk of about 8.6%, FDA reviewers wrote. (The difference was slightly higher when other methods of calculation were used.)
“Despite the modest treatment effect over placebo, the FDA cannot justify OCA use in NASH patients with Stage 2 or 3 fibrosis,” the reviewers wrote, referring to the level of internal scarring in patients’ livers at the time of treatment. The FDA estimates 5.7 million Americans fit that description and would be eligible for treatment with OCA.
”We've seen companies come back from negative docs with a positive vote but this is going to be a tough one to pull through,” wrote Baird analyst Brian Skorney in a note to clients on Wednesday.
Citing public comments from the company, Jefferies analyst Michael Yee wrote in a separate note that there’s a “chance” Intercept could discontinue development in NASH if OCA is rejected. The drug is sold as Ocaliva for a different liver disease known as primary biliary cholangitis, and generated about $343 million in worldwide sales last year.
No drug therapies currently exist for NASH, a disease caused by a buildup of fat in the liver and only slowed by weight loss or bariatric surgery. The disease affects 5% of the U.S. population and is the second-leading cause of liver transplants in the country, according to the FDA.
Drugmakers have pursued NASH drugs for more than a decade, but it’s proved a tough target. Several would-be medicines have failed in clinical testing. Intercept’s was the first to succeed, but regulators rejected it in 2020 and demanded more evidence.
At the time of OCA’s rejection, Intercept said FDA staff weren’t convinced OCA’s benefits outweighed its potential risks. Intercept spent the next few years collecting biopsies from more patients and resubmitted an application in 2022.
Along the way, though, shares in the company lost much of their value and newer competitors emerged. Madrigal Pharmaceuticals reported success in Phase 3 testing last year, for example. Others have disclosed promising results in earlier trials, too.
Wall Street analysts are therefore paying attention to Intercept’s review because of the potential implications for NASH drug development more broadly. According to Raymond James analyst Steven Seedhouse, the impact is likely to be limited. Still, he wrote in a Wednesday note that it would be “precedent setting for the entire field” if the committee votes to defer an approval until Intercept collects health outcome data.
“It’s hard to see the FDA saying wait for outcomes in this case, and then approving other drugs with similar ‘modest’ effects on surrogate endpoints unless safety is eminently clear,” Seedhouse wrote.
Jefferies analyst Yee added that others “may find negative FDA read-through to NASH,” given the harsh tone of the briefing documents. But he called the safety issues cited by the FDA specific to Intercept and not others with potentially safer or more potent drugs.
Intercept shares fell by as much as 20% before settling back to trade down about 15%. Shares in Madrigal fell by nearly 5%, while those of other NASH drugmakers Viking Therapeutics and Akero Therapeutics were little changed.