Most medicinal chemists design novel active pharmaceutical ingredients (APIs) to secure a patent and IP protection, not to create a scalable production process. That makes sense. They are racing against the clock, running through failures before they achieve success. Fine-tuning comes later.
When the technology is transferred into a production environment, a synthesis proven in the laboratory often doesn’t work at full scale. The synthesis route can be subject to process failures, or the process may not be efficient at commercial scale.
If you work with a contract development and manufacturing organization (CDMO), you need to know they can safely scale up your process and manufacture your compound efficiently. That work starts at tech transfer.
Get an early start
Finding obstacles early and addressing them methodically, as opposed to back-end troubleshooting, helps projects come in on schedule. Your CDMO should transfer in your technology “as is,” ideally early in Phase 2, and run the process as originally designed at lab scale. This allows the team – including analytical chemists, organic chemists, and production engineers – to obtain a baseline against which improvements can be measured. More important, it sets the stage for effective problem-solving.
Find scale-up challenges
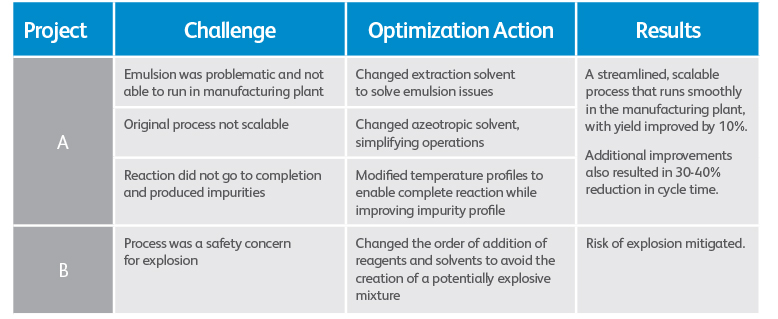
As the team gets familiar with your API’s process and chemistry, they identify issues that would occur at larger scale within the context of their plant’s equipment. An emulsion, for example, may behave differently in a shake flask versus a 150,000-liter tank. Or risk of explosion, which may pose minimal danger in the lab due to the small amounts of material involved, could be deadly at commercial scale.
Your CDMO should partner with your technical team to assess and, if necessary, redesign the production process based on their lab-scale assessments. Designed correctly for a cGMP manufacturing environment, the improved process will solve most problems associated with chemistry and manufacturing failures. But it shouldn’t stop there. You want efficiency, too, for sustainability over time and to improve your API’s economics, yield and supply assurance.
Optimize reaction conditions
Time is money when manufacturing API. The longer your process takes, the more it costs: time to prepare reagents, time in tank, time on filter, time to transport the intermediate to the next reaction. If you can squeeze out inefficiencies, you can glean savings at full-scale production.
To do that, you need to determine the boundaries of your critical process parameters. If your original process dictated running a reaction at 35 degrees, could you get better results at 34 degrees? Thirty-six? To dry your material, is heat alone best? Or would heat plus nitrogen be better? Do you need to dry your material to one percent before moving to the next reaction, per your original process? Or is five percent sufficient? These are the types of questions your CDMO team should be asking and testing at lab and/or pilot scale – before moving your molecule into commercial production.
Examples:
What to ask in your RFP
Don’t leave optimization to chance. In your Request for Proposal to potential CDMOs, let your CDMO candidates know you need them to optimize your API synthesis for both scalability and manufacturing efficiency. Ask how they generally approach these challenges. Also ask how they would partner with your technical team. A CDMO, no matter how skillful, cannot successfully optimize your process without side-by-side collaboration with you.
The bottom line is that you don’t want a CDMO to just take your process “as is” and run with it. Investing the time to optimize your synthesis process at lab scale for large-scale manufacture will yield you significant dividends over the long term.










