Tapping into a ubiquitous trend among businesses, one major CRO lab is developing an app to communicate more efficiently with its clients, investigators and clinical trial managers.
The goal of PPD Laboratories’ new app is to make the delivery of central lab data more useable, digestible and convenient, said Chris Clendening, executive director of project management and design for PPD Laboratories’ central lab. “We want to provide value to our customers who run clinical trials.”
The app takes to the mobile community what PPD Laboratories’ central lab provides through its new Preclarus® investigator site portal — a web-based platform that helps clinical trial sites manage their studies. Using a tech application familiar to mobile device users, investigators, doctors, trial coordinators and others can access study information in real time on their phones, laptops or tablets without having to log onto a fixed terminal, according to Clendening. The app, which will be available by the end of the year, will use the most current technology, including fingerprint recognition software to verify the user.
Combining the new app with PPD Laboratories’ in-house technology and lab management capabilities enhances the company’s offering, said David Stein, an independent eClinical consultant.
With a few screen taps, app users can carry out most of their frequent tasks much more quickly than having to navigate a larger system, he said. “It’s definitely the trend for vendors and CROs to create mobile apps. It makes a lot of sense for our industry to capitalize on the advantages that mobile solutions offer.” He said apps generally lead to greater productivity and efficiency, which ultimately cut costs.
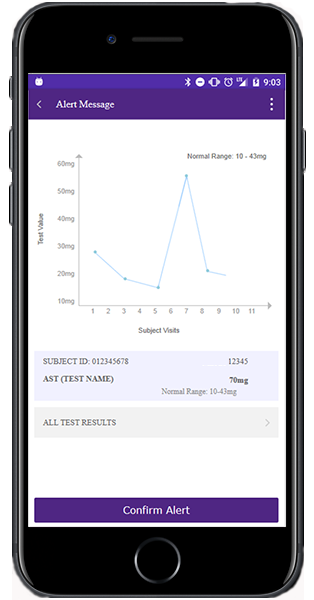
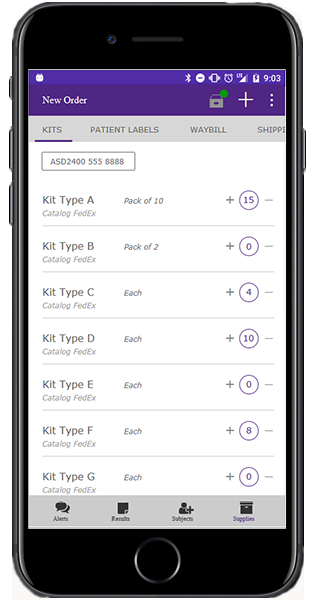
Virtual biorepository keeps track of patient samples
Building on the success of the Preclarus investigator site portal launched last year, PPD Laboratories is also expanding its lineup of clinical trial management tools. This includes customized portals for sponsors and third-party vendors, and a “virtual biorepository” for improved sample tracking. All of these enhancements should be available in early 2018.
It’s especially important to improve communication between the central lab and its users because of the sheer number of biomarker and genetic samples flooding the pharmaceutical industry, Clendening said. As the industry struggles to keep track of these samples, PPD Laboratories’ central lab is providing clients and third-party vendors new portals to the Preclarus central lab database with direct access to the sample data.
The virtual biorepository will provide visibility and access to stored samples and will give clients the ability to better monitor chain of custody, according to Brent Reed, PPD Laboratories’ associate director of IT, who is leading the development of the new tools.
PPD Laboratories’ clients and investigators also will be able to integrate their sample data more effectively, see where the samples are at any time, move them around as needed, order additional testing, keep track of other logistics, and easily upload test results for access by all parties involved in the clinical research.
Through new application programming interface (API) technology, many of these functions will be available for direct integration with partner information systems. This will result in an automated communication option, which will help increase data flows and decrease dependency on manual processes and procedures.
“Our experience at PPD is very significant because we are handling millions of samples across the world for our clients’ clinical trials, and must do so with a dedication to quality, accuracy and precision,” said Hacene Mekerri, vice president of operations for PPD Laboratories’ central lab.
Personalized landing pages allow easy access to custom and saved searches
In today’s connected world, especially in the clinical trials industry, security and authorized access to data is not just important, it’s imperative. Employing best practices, PPD is diligently working to improve the user experience while ensuring the data is always protected and always kept private.
Users of the new Preclarus portals will be directed to their own landing page based on their unique roles. Their view will be tailored to their usage, without having to navigate deeply to find the data and reports they routinely access, Reed said.
Leveraging advanced report-building technology, users will be able to design their own custom queries and real-time searches on data that PPD Laboratories produce rather than having to rely on the company’s data management teams to design such reports, he said.
“That’s a game changer. That’s providing real-time access to data. That’s critical to bending the cost and time curve not only for our company, but for our customers. At the end of the day, the data we produce contributes to the approval or disapproval of a drug. If we can provide tools to our customers to analyze data as a trial progresses, it enables them to make operational adjustments that strengthen the likelihood of success when the drug is submitted to regulatory bodies,” Reed said.
According to Clinton White, vice president of enterprise information management, trial sponsors will have more accessible, usable and relevant data at their fingertips, enabling them to make better, more-informed decisions and ideally cutting costs and speeding the drug-to-market timeline. “We try to make sure we automate and streamline,” he said. “We are trying to present the right data as quickly and clearly as possible … at the right time to the right user’s hand to make the right decision.”
With an eye to the future, PPD Laboratories continues to look for innovative ways to improve the clinical drug delivery process and enable its clients to be more successful, White said. Its new app and advanced communication tools are bringing clinical trial managers, sponsors and investigators closer to that goal.










