Selecting the right primary container for an injectable drug product is a crucial step in the drug development process. A wrong choice is likely to cost significant time and resources later in the process, as well as potentially cause regulatory issues. So, the pressure is on to get it 'right the first time'.
Drug stability and sensitivity will affect the type of container that is appropriate. There are also container-specific factors to take into account – including cosmetic appearance, filling equipment, chemical durability and container closure integrity. Manufacturability must also be considered, as well as long-term lifecycle management – if a drug introduced in vial format may eventually be switched to a pre-fillable syringe, for example, due to market preferences.
A proper risk assessment early in the product development process can mitigate these issues and help to save time and money. That's where a drug containment solutions partner can play a vital role – providing guidance and risk analysis for both short-term and long-term lifecycle management. With more than 70 years of experience in the production of advanced glass containment solutions for the biopharmaceutical industry, Stevanato Group is ideally placed to rise to the challenge.
The COVID-19 pandemic illustrated the vital importance of getting things right the first time when it comes to drug development and delivery. The pandemic saw the first large-scale use of mRNA technology for vaccines, with new challenges thrown up by the extreme cold-storage conditions the vaccines required.
Primary packaging suppliers and pharma companies were faced with limited data and experience at such low temperatures, particularly in terms of container closure integrity and the mechanical performance of the containers.
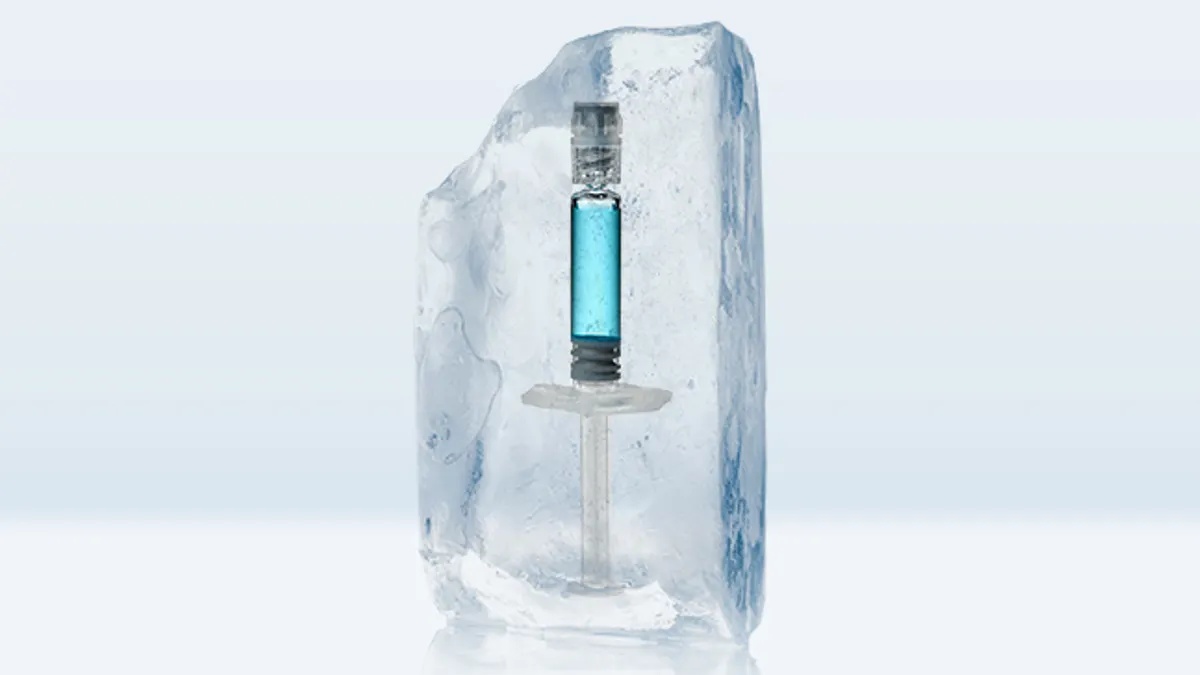
Container closure integrity was one of the biggest challenges to overcome especially for syringes, as they are far more complex than vials. The concern was whether there would be any risk of loss of sterility during the freezing and thawing cycles.
In a syringe, the expansion of water-based liquids when frozen results in a lateral force moving the plunger towards the flange. Stevanato Group's EZ-Fill® glass syringe with an integrated tip cap is designed to overcome this problem. The rubber tip cap is mechanically fixed in a plastic holder and prevents leakage by securely closing the front part of the syringe.
Contraction of air during the freezing process also results in plunger movement, particularly in syringes that are empty (air filled). Stevanato Group's recommendation was therefore to adopt an airless filling process, if possible – or at least keep the amount of air between the drug and the stopper to a minimum.
Plunger stopper shrinkage and changes in material properties at very low temperatures can also affect seal integrity. The mismatch of the coefficients of thermal expansion of glass versus rubber can cause a different shrinkage at low temperatures. In addition, the rubber exhibits a glass transition temperature close to the target temperature that could change the mechanical properties from a viscous/rubbery state to a more hard and relatively brittle one.
As well as container closure integrity testing, Stevanato Group scientists also investigated the impact of deep freezing on other critical parameters such as mechanical resistance, break-loose and glide force, plunger glide force and the torque required to unscrew the integrated tip cap. Extensive testing by the company's analytical services teams confirmed the suitability of SG EZ-Fill® ITC glass syringes for drugs requiring storage at very low temperatures.
With the use of mRNA technology likely to extend beyond vaccines to cancer treatment and other therapies in the future, choosing the right drug containment solution has never been more important.