Editor’s note: BioPharma Dive, as part of its newly launched Emerging Biotech Weekly, is taking a closer look at competitive areas of startup activity. We aim to give an overview of the companies developing a new technology and what their goals are. This, on in vivo cell therapy, is our sixth.
In 2011, cancer cell therapy was closer to science project than treatment.
Immunologist Carl June and his colleagues at the University of Pennsylvania were struggling to raise money for a small study of a cell therapy for leukemia. The trial “was an academic exercise,” said June. “We had no idea it would turn into a commercially viable product.”
The treatment June was working on, now sold as Kymriah, became the first CAR-T cancer therapy to reach market, establishing cell therapy as a new class of medicines. Other, similar treatments have followed since and are used to treat leukemia, lymphoma and multiple myeloma. When they work, they can produce durable remissions.
Yet, despite a decade of rapid progress since June’s 2011 study, CAR-T therapy’s reach remains limited. It hasn’t been used effectively for non-malignant diseases and, even in oncology, its success is confined to blood cancers.
Many companies are working to overcome CAR-T’s shortcomings and broaden its potential applications. One strategy involves turning CAR-T from a complex process performed outside the body into a simpler infusion. This inside-the-body, or “in vivo,” approach could make cell therapy more accessible. It also may open up other conditions for CAR-T treatment.
At least five startups have formed in recent years to develop in vivo cell therapy. June co-founded one of them, Capstan Therapeutics, and the rest have drawn interest from top-tier investors or large pharmaceutical companies. Here’s where things stand:
What is in vivo cell therapy and how does it work?
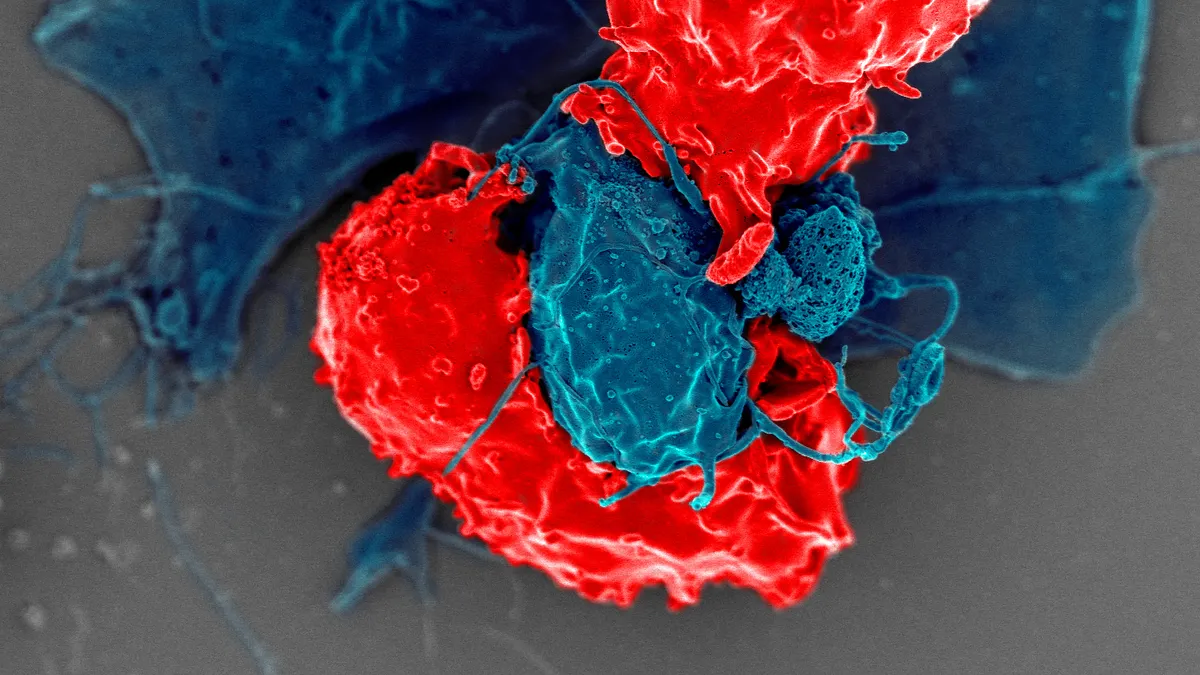
Constructing a CAR-T therapy is delicate, laborious work. Doctors must first draw out a patient’s white blood cells, which are then frozen and shipped to a laboratory. There, scientists alter T cells by adding a gene for a new receptor — called a chimeric antigen receptor, or CAR — that helps them grab proteins on the surface of cancerous cells. These souped-up T cells are multiplied many times over, frozen and — about two to three weeks after the whole process began — reinfused back into the patient.
With Kymriah, for example, T cells are modified to lock onto a protein called CD19 that’s expressed on cancerous B cells in leukemia and lymphoma. Multiple myeloma cell therapies like Bristol Myers Squibb’s Abecma, by comparison, consist of T cell hunters seeking a protein called BCMA.
Once these engineered CAR-T cells are infused, they search for and destroy cancer cells expressing their target proteins, sometimes driving an immune response so strong it can be life-threatening.
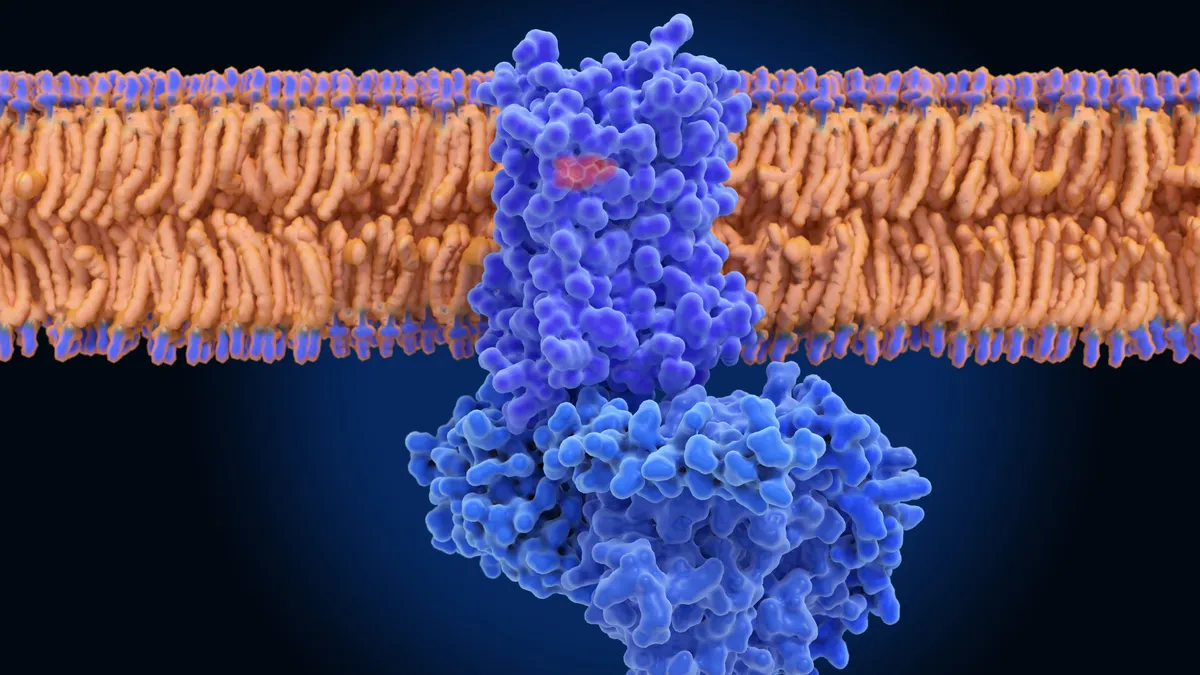
As the bulk of this work takes place in a lab, CAR-T is considered an “ex vivo,” or outside-the-body, procedure. With in vivo cell therapy, companies aim to modify immune cells inside the body, with the help of technologies like gene editing and messenger RNA.
June’s startup, Capstan, is using mRNA technology similar to that involved in COVID-19 vaccines to teach T cells to recognize diseased cells. Its founders showed that mRNA stuffed into fatty spheres known as lipid nanoparticles are taken up by heart cells in mice, where they were translated into new surface proteins and helped repair damaged tissue. Umoja Biopharma, meanwhile, is using lentiviruses to deliver genes designed to help the body make its own CAR-T cells.
Other startups are also turning to in vivo cell engineering. Ensoma is carving out the viral genome of an adenovirus and using it to carry gene editing instructions into the body. Vector BioPharma, meanwhile, uses virus-like particles to transfer genetic material into the body. Both approaches could be used to treat inherited diseases as well.
These methods come with risks. Synthetic mRNA, for example, can trigger an immune response that, while helpful for a vaccine, could be harmful when used as treatment. Adenoviruses have a checkered past, having been the source of safety concerns in gene therapy experiments long ago. Lentiviruses can erroneously trigger cancer-causing mutations, a risk flagged by the Food and Drug Administration in the review of two Bluebird bio gene therapies and a reason they’re mostly involved in tightly controlled ex vivo processes.
What advantages does in vivo cell therapy have over existing treatments?
CAR-T therapies take weeks to produce. While patients wait, their disease can worsen or they might become too weak to receive treatment. They also require a “conditioning” chemotherapy regimen to prepare them for treatment, and monitoring for weeks afterwards to guard against potentially severe side effects.
These hurdles have made CAR-T an expensive treatment primarily available at specialized centers and less accessible to people who live in rural areas or poorer nations. They’ve also weighed on the commercial outlook of other “ex vivo” gene-based treatments for inherited diseases like sickle cell and beta thalassemia, which are made with similarly complex manufacturing processes.
An “in vivo” solution delivered through an infusion could be quicker and less costly to manufacture. Inside-the-body techniques could also provide an alternative to the “off-the-shelf” therapies involving donor cells, which have safety concerns and efficacy questions of their own. They might eliminate the need for chemotherapy conditioning regimens.
Some companies working on inside-the-body approaches aim to overcome the limitations of gene therapies that rely on adeno-associated viruses, or AAVs, to deliver their genetic cargo. While AAVs are widely used, the effects of the treatments they deliver may wane in cells that frequently divide. They can only carry a small amount of genetic material, too, and are sometimes targeted by the body’s defenses.
What companies are working on in vivo cell therapy?
At least five startup companies have emerged since last year with technologies they claim are capable of producing in vivo cell therapies.
Umoja was seeded by MPM Capital and DCVC Bio and formed by scientific researchers at Seattle Children’s Research Institute and Purdue University. It has since raised more than $260 million and brought its first drug candidate into clinical testing in May.
Ensoma officially launched in February 2021 alongside a wide-ranging partnership with Takeda that covers up to five experimental programs. It’s run by Emile Nuwaysir, the former CEO of Bluerock Therapeutics, a cell therapy startup Bayer bought in 2019.
Recent startups developing in vivo cell therapies
| Company | Top investors | Total raised to date |
|---|---|---|
| Umoja BioPharma | MPM Capital, DCVC, SoftBank Vision Fund, Comorant Asset Management | $263M |
| Capstan Therapeutics | Novartis, RA Capital, OrbiMed, Pfizer, Bayer, Eli Lilly, Bristol Myers Squibb | $165M |
| Interius BioTherapeutics | Comorant Asset Management, Fairmount Funds, Bain Capital, Pfizer | $76M |
| Ensoma | 5AM Ventures, Takeda, F-Prime Capital | $70M |
| Vector BioPharma | Versant Ventures | $30M |
SOURCE: Companies
Interius BioTherapeutics followed in May 2021 with $76 million from Cormorant Asset Management and others including Bain Capital and Pfizer’s venture arm. The company is based on research by UPenn physician-scientist Saar Gill and is led by Phil Johnson, who was once chief scientific officer at The Children’s Hospital of Philadelphia.
Vector BioPharma launched in August with $30 million from Versant Ventures and technology spun out of protein engineering expert Andreas Pluckthun’s lab at the University of Zurich. The month after, Capstan, co-founded by June, fellow CAR-T innovator Bruce Levine and mRNA pioneers Drew Weissman and Hamideh Parhiz, came out of stealth with the backing of five pharma companies.
Publicly traded biotech Sana Biotechnology is also working on in vivo cell therapy. Novartis, meanwhile, is developing a CAR-T manufacturing process that minimizes the amount of work done outside the body, allowing modified cells to expand inside patients rather than in a lab.
What is the status of their research?
Most in vivo cell therapy work is still in early stages, though some developers have revealed details about their plans.
Sana has more than half a dozen experimental treatments in preclinical testing for blood cancers and rare inherited diseases. It expects to ask the FDA later this year to begin Phase 1 testing of one candidate being developed for two types of leukemia and non-Hodgkin lymphoma.
Umoja’s first program, for a type of bone cancer tumors, is a typical ex vivo CAR-T treatment with a separately administered drug that “tags” tumor cells for destruction. The company could ask regulators as soon as next year to begin human testing of two in vivo programs for solid tumors and blood cancers, according to its website.
Vector expects to have “technical proof-of-concept” data in 2022 and animal data for its lead programs in 2023, it said in a statement.
Capstan is starting with in vivo CAR-T therapies for diseases with no effective treatments, but plans to develop drugs for certain genetic blood disorders too. It hasn’t disclosed timelines. Neither have Ensoma or Interius, the latter of which is also looking at cancer immunotherapy.