Merck & Co.’s $6.7 billion bid for Terns Pharmaceuticals took Wall Street by surprise, both because of its timing and the lower-than-anticipated price being paid for a much-heralded treatment that could reshape treatment of chronic myeloid leukemia.
Terns’ drug, called TERN-701, is a type of targeted, oral treatment that’s shown early potential to challenge marketed therapies, like Novartis’ Scemblix, in the treatment of CML. Yet Wall Street analysts have forecast peak annual sales estimates that effectively surpass the net $5.7 billion Merck is paying, when accounting for Terns’ cash reserves.
The 6% purchase premium, among the lowest paid for a publicly traded drugmaker since at least 2018, according to BioPharma Dive data, also added to a sense by some analysts and investors that rival companies may swoop in and top Merck’s offer.
Moreover, investors dissatisfied with the deal terms could refuse to tender their shares because they view the Merck offer as undervaluing the company.
“In our assessment, this deal price vastly underestimates the potential for TERN-701,” wrote Leerink Partners analyst Andrew Berens, who covers Terns, in a note to clients. A different buyer paying a “reasonable multiple” of 1.5 to two times peak forecasted sales would represent a “meaningful premium to the current bid,” he wrote.
Terns was formed as a cross-border company aiming to bring innovative medicines to China and initially focused on a common liver disorder called metabolic dysfunction-associated steatohepatitis. The company licensed treatments from Eli Lilly and raised $128 million in an initial public offering in 2021 to support development. Shares premiered at $17 apiece.
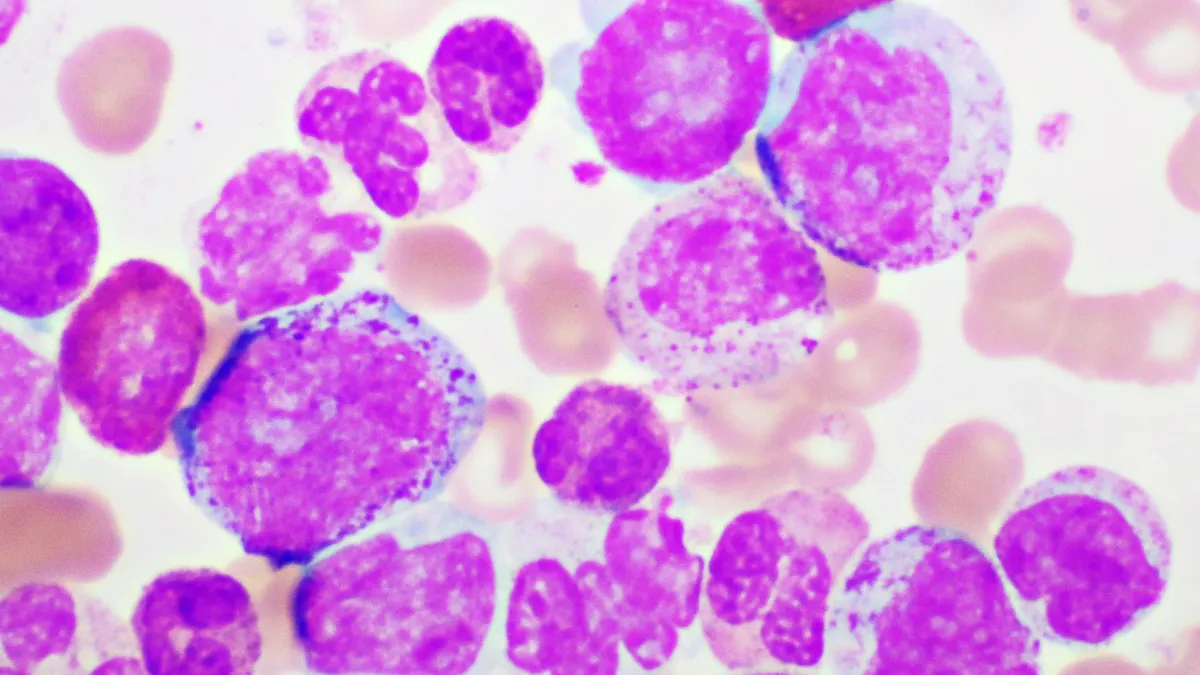
Terns’ work in MASH, and then later, obesity, was eventually sidelined, sinking shares as low as less than $2 apiece at one point and under $3 as recently as a year ago. In the meantime, though, it had another therapy, licensed from China’s Hansoh Pharmaceutical in 2020, that was progressing against CML. That therapy, now known as TERN-701, moved to the forefront as Terns’ other research struggled and has been the source of a stock revival.
The therapy’s potential was showcased at the American Society of Hematology meeting in December. Data Terns disclosed from an early-stage trial showed a majority of people whose chronic myeloid leukemia had progressed on other similar drugs saw a “major molecular response,” or a vast reduction in the number of diseased blood cells, after being treated with TERN-701. Analysts were so impressed by the findings they began suggesting Terns’ drug upend care for CML.
Terns shares rocketed to more than $47 apiece following the meeting and have steadily climbed in early 2026. The company’s stock price closed at $50 on Tuesday, the day before Merck announced that Terns had accepted a $53-per-share bid.
On a conference call with analysts Wednesday, Merck CFO Caroline Litchfield said the deal’s terms capture the value of TERN-701’s sales potential in the U.S. and assume no pricing restrictions from Medicare as well as market exclusivity that extends into the 2040’s.
To some analysts, that price represents a fair deal. Given Terns’ long-depressed stock price, the deal “represents an excellent outcome for [Terns] shareholders,” Mizuho Securities analyst Graig Suvannavejh wrote in a note to clients.
Others are convinced Terns is being undervalued. Berens, of Leerink, projects that if TERN-701 is approved, it’ll surpass $1 billion in annual sales by 2032 and peak at $6.2 billion yearly in 2040. Most people with CML survive 10 years or more following their diagnosis and can expect to have a normal lifespan, making it likely that a tolerable drug that keeps the disease in check will be a big seller. Terns’ drug, then, should be able to generate “multi-blockbuster revenue,” especially if it becomes the treatment standard for newly diagnosed patients, Berens wrote.
For Merck, those ambitious projections could help it offset the revenue decline expected when its biggest seller, Keytruda, loses patent protection. Merck is eyeing $70 billion in annual sales next decade, and has looked to a combination of acquisitions and in-house research progress to reach that target.
The price it negotiated for Terns “represents shrewd decision-making” by Merck’s business development team, RBC Capital Markets’ Trung Huynh wrote, adding that the absence of non-guaranteed, success-based payouts suggests Merck bested other competitors with a winning bid.
Many investors are criticizing rival large drugmakers “for being asleep at the wheel allowing such a valuable asset to be acquired at a discount,” Hyunh wrote. Yet he and others suggested companies like AbbVie or Bristol Myers Squibb might jump in with a competing offer.
“We believe any large biopharma with an interest in establishing or building its presence in oncology would be interested,” Suvannavejh wrote.