Editor’s note: BioPharma Dive, as part of its newly launched Emerging Biotech Weekly, is taking a closer look at competitive areas of startup activity. We aim to give an overview of the companies developing a new technology and what their goals are. This, on TYK2 inhibitors, is our fifth.
When one drugmaker succeeds, many others try to do better.
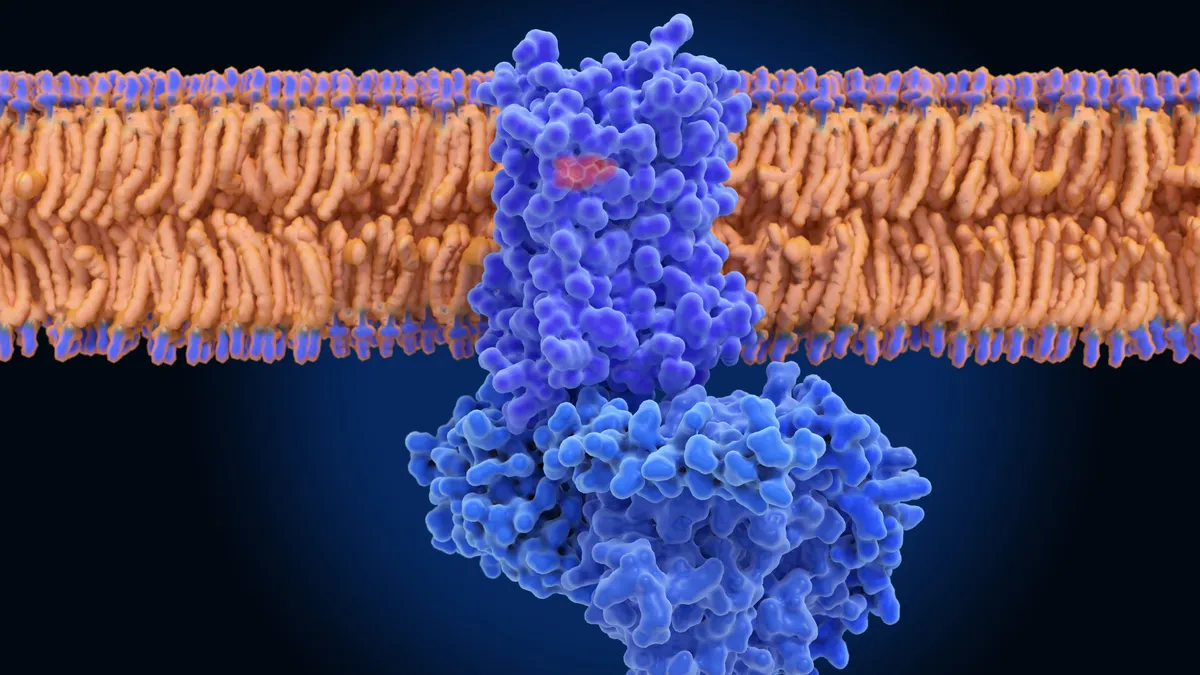
That’s one reason why a protein called TYK2 has fast become one of the biotechnology industry’s top targets. At least a half dozen startups have set out to develop drugs that block it to better treat autoimmune diseases.
Conditions like psoriasis and arthritis affect millions of people. For years, they have been treated with injectable drugs like AbbVie’s Humira, which are among the sector’s most lucrative products.
Drugmakers have sought to replace them with more convenient oral medicines, but with mixed results. Pfizer, AbbVie and Eli Lilly, for example, each brought to market pills that target a family of proteins known as Janus kinases, or JAKs, which are involved in the body’s immune response. The drugs have proven effective at treating a wide range of inflammatory diseases, yet their use has been curtailed due to safety concerns.
Enter the TYK2, or tyrosine kinase 2 protein. Though part of the broader JAK family, TYK2 is viewed as a potentially safer target by scientists and drugmakers. That thinking was reinforced in September, when the Food and Drug Administration approved Bristol Myers Squibb’s Sotyktu for moderate-to-severe psoriasis without the safety warnings that have limited its JAK-blocking competitors.
Now the race is on to surpass Sotyktu. At least one startup developing a TYK2 inhibitor has launched since Sotyktu’s approval, while two other companies raised fresh funding to push their drugs further along.
Success could mean significant financial gain. Bristol Myers, which is also testing Sotyktu in psoriatic arthritis, lupus, and Crohn’s disease, forecasts yearly sales of Sotyktu could eventually reach as high as $4 billion.
Here’s where things stand.
What are TYK2 inhibitors and how do they work?
TYK2 inhibitors work by blocking the TYK2 protein and the cellular signals that run through it. Those signals can in turn activate other immune proteins and are associated with inflammation.
Disrupting that signaling by targeting TYK2 is viewed as a promising way to tamp down excessive inflammation brought on by autoimmune diseases such as psoriasis, psoriatic arthritis, Crohn’s disease and lupus.
Another part of the attraction of TYK2 inhibitors is they offer a way to target inflammation without also blocking related JAK proteins, the inhibition of which can cause serious side effects.
The first approved JAK inhibitor, known as Jakafi, was discovered by biotech company Incyte and developed for myelofibrosis and graft-versus-host disease. Since then, drugmakers have poured hundreds of millions of dollars into development of JAK blockers for autoimmune conditions, leading to Pfizer’s Xeljanz, AbbVie’s Rinvoq and Eli Lilly’s Olumiant. All three are approved for rheumatoid arthritis, as well as conditions like psoriatic arthritis that are being targeted by TYK2 developers.
But while JAK inhibitors intrigued researchers because of their ability to block multiple inflammatory cytokines, that kind of broad effect has also been their weakness.
What advantages do TYK2 inhibitors have over other treatments?
So far, TYK2 inhibitors haven’t been linked to the serious health problems that have been reported in testing of JAK inhibitors.
In clinical studies, some patients taking the JAK inhibitors developed by Pfizer, AbbVie and Eli Lilly developed infections and blood clots, meriting an FDA warning on the drug’s labeling. Additionally, a study comparing Pfizer’s Xeljanz to drugs like Humira showed a higher rate of cardiovascular complications and cancer. That finding led to the FDA to require JAK inhibitors be used only in patients who cannot take or don’t respond to those drugs.
While infections were observed in testing of Sotyktu, the majority weren’t serious and did not lead to patients discontinuing treatment. The FDA didn’t include boxed warnings of the risk of clotting, cancer or heart complications on Sotyktu’s labeling, either, despite some concerns the agency might consider anti-TYK2 drugs as similar to JAK inhibitors.
Doctors can also prescribe Sotyktu without requiring patients first try biologic drugs. In testing, Sotyktu bested Amgen’s Otezla, an oral treatment for psoriasis that works differently, indicating TYK2 blockers can be potent options for clearing skin, too.
As pills, TYK2 blockers are also more convenient than injectable drugs, such as Humira or newer medicines like Novartis’ Cosentyx, which are widely prescribed.
Which companies are developing TYK2 inhibitors?
Aside from Bristol Myers and Galapagos, another large drugmaker working on a TYK2 inhibitor, a handful of startups have cropped up to break into the field.
One of the splashiest is Priovant Therapeutics, a startup spun out of Pfizer and Roivant Sciences in June with two TYK2-targeting candidates. Pfizer holds a 25% stake in Priovant, which has not raised additional funding.
The most well-funded, Nimbus Therapeutics, has raised at least $400 million in private financing for development of its TYK2 inhibitor and other drug candidates, according to the company. Adding in partnerships and other deals, the company has to date received more than $1 billion, Nimbus company CEO Jeb Keiper recently told BioPharma Dive.
Select list of biotech startups developing TYK2 inhibitors
| Company | Lead Investors | Amount Raised |
|---|---|---|
| Nimbus Therapeutics | RA Capital Management, BVF Partners and Atlas | At least $400 million |
| Alumis Therapeutics | Foresite Capital, AyurMaya | $270 million |
| Neuron23 | Westlake Village BioPartners, Kleiner Perkins, Redmile Group, SoftBank | $213.5 million |
| Sudo Biosciences | Frazier Life Sciences, Velosity Capital | $37 million |
SOURCE: Companies
Another company, Alumis, is working on a TYK2 inhibitor as well. Formerly known as Esker Therapeutics, the company raised a Series B round in January that brought its funding total to $270 million.
Ventyx Biosciences went public in 2021 with a $152 million initial stock offering, and in September added another $177 million in financing.
Sudo Biosciences is the latest company to launch, emerging from stealth two weeks after Sotyktu reached market.
What is the status of their research?
The clean label given to Sotyktu has brightened the prospects of would-be competitors.
Furthest along in testing is Priovant, whose lead candidate, brepocitinib, blocks both TYK2 and JAK1. Unlike Bristol Myers and other startups, Priovant isn’t focusing on psoriasis. The company started a Phase 3 study in the spring testing brepocitinib for a rare skin and muscle disorder called dermatomyositis, and is evaluating the drug in lupus as well.
Alumis, meanwhile, began a Phase 2 trial of its drug in plaque psoriasis in late September and said it plans to start more mid-stage studies “in the near future.”
Galapagos has reported results from a Phase 1 trial of its TYK2 inhibitor in psoriasis, and a Phase 2 trial in dermatomyositis is expected to start by the end of 2022. A rival psoriasis drug developed by Ventyx has also read out Phase 1 results.
Other companies are nearing the clinic. Startup Neuron23 is working on a drug candidate that it expects to push into preclinical studies by early 2023, the company said.
Ventyx, Sudo and Nimbus have each claimed their drugs could be more selective and potentially more effective than Sotyktu. They have a ways to go before they can prove that claim with clinical trial data, however.