Dive Brief:
- The Food and Drug Administration will allow Beam Therapeutics to proceed with the first clinical trial testing a new type of gene editing, the high-profile biotech company announced Monday, in a milestone for a field that's advanced rapidly over the past several years.
- The Phase 1/2 study will test BEAM-101, a therapy designed to treat sickle cell disease by reactivating a form of the oxygen-carrying protein hemoglobin that normally disappears soon after birth. While the approach is shared by other gene editing biotechs working on sickle cell treatments, Beam believes its "base editing" technology can be safer and more precise.
- Alongside the clinical trial milestone, Beam also announced that John Maraganore, Alnylam Pharmaceuticals' longtime CEO, will join its board of directors. Beam is at "an important inflection point," Maraganore, who's soon to step down from his post at Alnylam, said in a Nov. 8 statement.
Dive Insight:
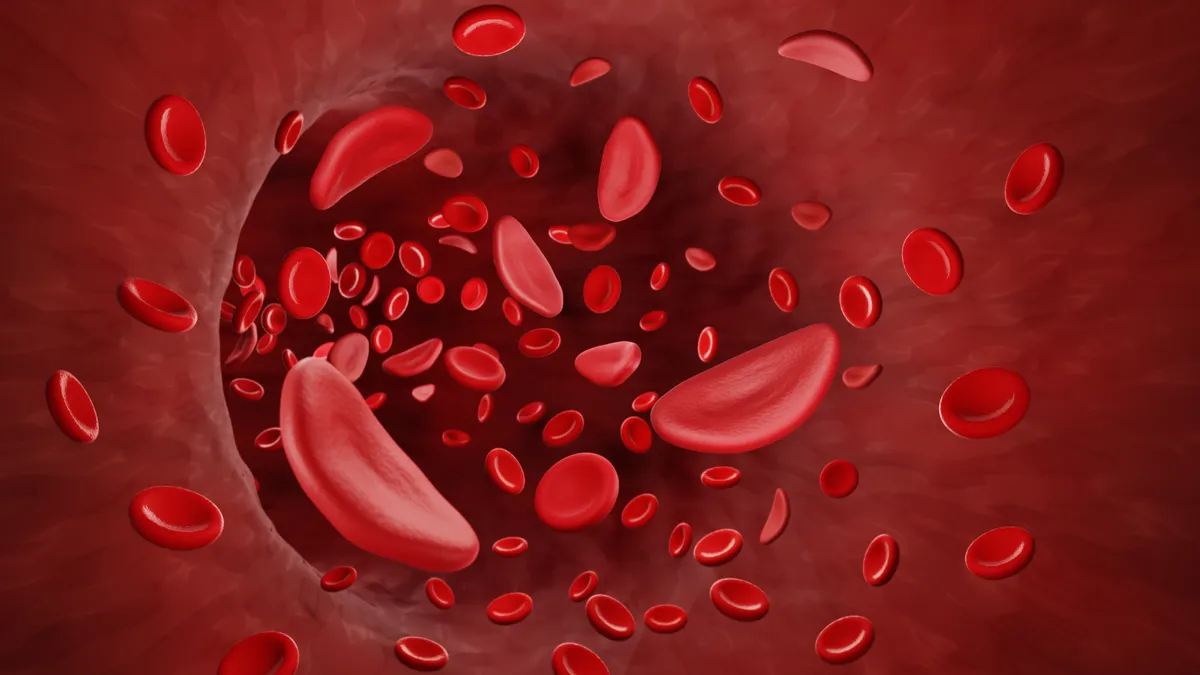
Gene editing's promise is clearest, and perhaps closest, in sickle cell disease, a hereditary condition characterized by deformed, or "sickled," red blood cells that cause painful blockages and damage organs.
A therapy being developed by CRISPR Therapeutics, one of the first biotech companies formed to advance the now famous gene editing technique, and Vertex Pharmaceuticals has produced dramatic results in just over half a dozen patients treated to date. After treatment with the companies' medicine, which uses CRISPR to edit stem cells extracted from each patient, none of seven experienced any of the pain crises. Data also suggested a long-lasting effect from the editing performed on cells.
The changes introduced by CRISPR and Vertex's treatment are designed to reactivate what's known as fetal hemoglobin, a form of the protein that's made in the first few months of life and then shut off afterwards. People with a condition in which production of fetal hemoglobin persists into adulthood are typically protected from sickle cell symptoms even when they carry mutations that would otherwise trigger the disease.
Beam's doing something similar with BEAM-101, but it's using a more precise form of gene editing than CRISPR. Base editing, as it's called, is designed to alter single DNA "letters" rather than larger strips of genetic code, and does so without breaking both strands of the DNA double helix. Other companies, including the heart disease-focused Verve Therapeutics, hope to use base editing to treat other conditions influenced by genetics.
Beam's trial will be the first test of the technology in humans and, as such, is an early proof point for how the principles of CRISPR gene editing can be expanded and advanced. Michael Yee, an analyst at Jefferies, wrote in a note to clients that he expects the first patient could be dosed sometime next spring with first data possible by the end of 2022.
If Beam succeeds, though, there's a good chance other gene-modifying treatments will be available for sickle cell disease. Along with CRISPR and Vertex's therapy, Bluebird bio is far along in testing a gene therapy for the blood disorder and other companies like Sangamo Therapeutics are exploring different types of gene editing, too.
Beam has acknowledged the competition, but believes its therapy can be safer through its hypothetically more precise editing. The company is also studying a second sickle cell treatment that uses base editing to directly correct the sickle cell mutation, which could offer other long-term benefits.
So far, Beam has capitalized on the promise surrounding its technology and the broader gene editing field, raising significant funds through private placements and an initial public offering last year. Proving its technology works in humans is a much harder task.
Having Maraganore, who successfully piloted Alnylam through the decades-long development of therapies that alter RNA expression, on the board could help. Investors, at least, seemed encouraged by the twin Monday morning updates, sending shares in Beam higher by nearly 8%.