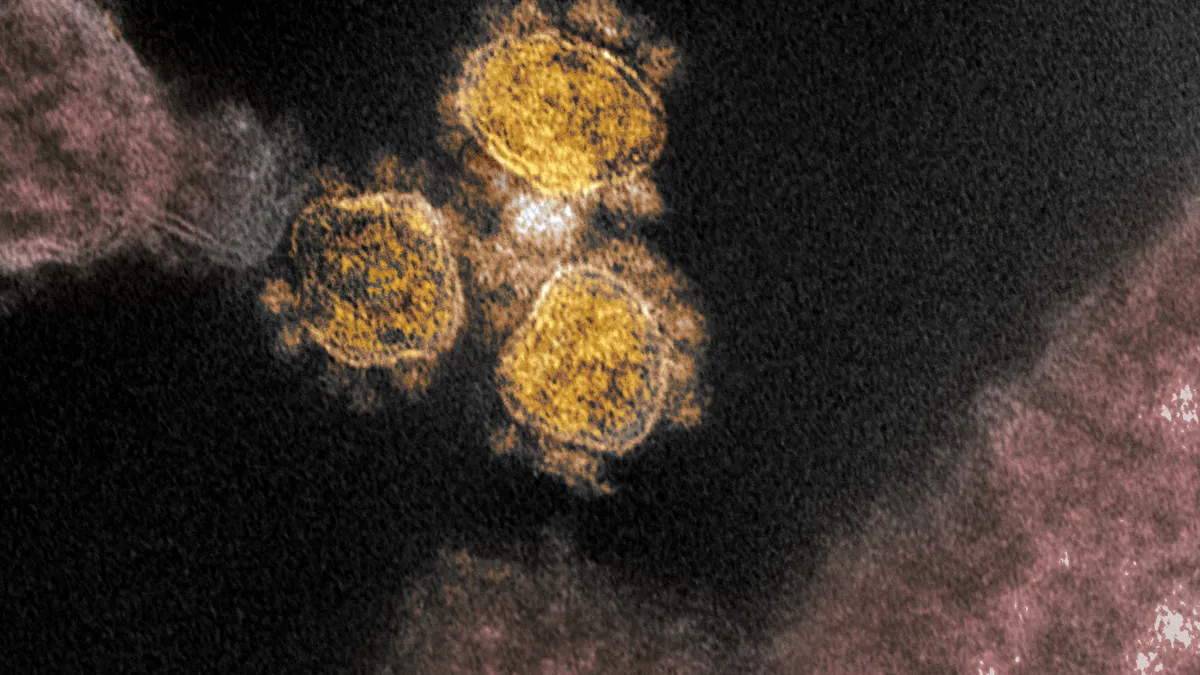
No medicines are proven to treat the new coronavirus which emerged from China late last year and has since jumped to more than 100 countries.
With little else but supportive care to check the virus' rapid spread, doctors and scientists have launched hundreds of trials in search of an effective therapy, testing mostly repurposed HIV or flu drugs.
Hopes are highest for remdesivir, an experimental antiviral developed by Gilead for use against Ebola but since pressed into study for the new coronavirus.
"There's only one drug right now that we think may have real efficacy. And that's remdesivir." said Bruce Aylward, a senior advisor and international leader of the World Health Organization's joint mission to China, at a Feb. 24 press conference.
Clinical trials are now underway in the U.S., China and, soon, other Asian countries where high numbers of people are infected by the virus, now named SARS-CoV-2. Preliminary results from the first of those studies could come by April, Gilead expects.
Other drugs designed specifically to treat SARS-CoV-2 are now in development but, for the next few months at least, much of the focus will be on remdesivir. Here's a primer on how it works, its potential against the illness COVID-19, as well as why it's no sure thing.
What kind of drug is remdesivir?
Remdesivir is designed to slow the infection of healthy cells by blocking viral replication, checking the ability of an invading virus to co-opt the body's cellular machinery to reproduce itself.
In technical terms, it's known as a nucleotide analog — meaning it mimics a genetic building block, but with an unusual molecular group tacked on. The thinking goes that, as the invading virus tries to replicate itself, the drug gets in the way and stops the process.
Remdesivir emerged from work done by Gilead and U.S. government in the mid-2010s to test compounds discovered by the biotech against emerging viruses. Initial animal experiments seemed to show the drug could work against Ebola, leading Gilead to start Phase 1 testing in 2015 and advance it into a mid-stage trial the next year.
A large Phase 3 study conducted in the Democratic Republic of the Congo, however, showed remdesivir to be less effective in preventing deaths from the virus than two other drugs, leaving Gilead's therapy with an uncertain future.
But the outbreak of SARS-CoV-2 made suddenly relevant a series of experiments in human cell cultures and mice that indicated remdesivir could have activity against coronaviruses like MERS and, possibly, the new one now spreading.
What evidence suggests remdesivir could treat COVID-19?
Gilead has taken pains to emphasize, when announcing new plans for remdesivir, that it lacks human data on whether the drug actually works to treat COVID-19.
What it does have is preclinical data showing the drug to be active against the MERS and SARS viruses, which share enough genetic similarities to SARS-COV-2 that researchers and the company think remdesvir could work against it too.
Testing by National Institutes of Health researchers, published in January, demonstrated remdesivir prevented MERS from developing in monkeys, and helped improve the condition of those animals that were already infected.
"Our results, together with replication inhibition by remdesivir of a wide range of coronaviruses in vitro and in vivo, may further indicate utility of remdesivir against the novel coronavirus [...] emerging from Wuhan, China," they wrote.
Most recently, an in vitro study conducted by scientists at the Wuhan Institute of Virology found remdesivir and an anti-malarial called chloroquine more likely to be effective against SARS-CoV-2 than a handful of other potential treatments.
As remdesivir has drawn attention, many have focused on its use in treating a Washington man who tested positive for SARS-CoV-2 after returning from Wuhan. One week into his hospitalization, doctors gave him remdesivir through a compassionate use program that Gilead has been supporting.
The man's condition began to improve the next day, sparking some optimism about remdesivir's benefit. But it's not clear if the patient had already begun to recover, as viral levels were declining before the drug's use.
How is remdesivir being tested now?
Early last month, doctors in Wuhan and Beijing opened two Phase 3 studies of remdesivir in coronavirus-infected patients with either mild to moderate illness or with severe disease. The former began enrolling on Feb. 6 and the latter a week later, on Feb. 13.
Together the trials aim to treat about 850 people and measure whether intravenous remdesivir can improve or resolve the symptoms of COVID-19 over the course of roughly a month.
Investigators and Gilead expect results by April, which would give an early signal of how likely the drug is to play a role in checking the fast-spreading epidemic. Yet enrollment has not been as smooth as might be expected. The WHO's Aylward noted last month that a surge in studies of other potential treatments has made recruiting more difficult, as have restrictions limiting who can join the two trials.
In the U.S., a study sponsored by the National Institutes of Allergy and Infectious Disease recently began recruiting patients at hospitals in Nebraska and Washington, with sites planned in Maryland and Texas. The first participant was an American repatriated from Japan after being quarantined on the Diamond Princess cruise ship in Yokohama harbor.
Other treatments could be added into the study in the future, which is designed to allow modifications over time.
Gilead, meanwhile, recently announced plans to start two of its own Phase 3 studies in Asian countries where there are high numbers of diagnosed cases. Those will enroll about 1,000 patients and begin "within days," Gilead CEO Daniel O'Day told reporters at a briefing in Washington, D.C. on Thursday.
"By the time we get to the end of April, we should have a preliminary idea of the safety and efficacy of this medicine against coronavirus," O'Day said.
How would remdesivir be used, if tests show a beneficial effect?
Despite the evidence suggesting remdesivir could work, it's still an open question whether that potential will translate into real efficacy. There was preclinical and early clinical data suggesting the drug would protect patients from Ebola, too, but results from that Phase 3 study still disappointed.
"It is too early to place much confidence in the compound's likely activity against this virus, and even if it does prove active, we would expect relatively modest activity in larger populations, rather than an immediate and potent response such as could be seen with an antibacterial," wrote analysts at SVB Leerink in a recent note to clients.
The drug's dosing could also limit use, should tests show it to be beneficial. The Phase 3 studies in China are using a 10-day course of intravenously infused remdesivir, while Gilead is testing a 5-day course in its trials.
Patients receiving the drug, therefore, would have to be in a hospital and would likely need to be experiencing more serious symptoms of COVID-19, such as pneumonia or shortness of breath.
And while remdesivir was used in hundreds of patients with Ebola, its safety and appropriate dose have not been studied for COVID-19 as much as typically would be the case with late-stage testing.
If remdesivir is proven, how would Gilead respond?
Gilead built up clinical supplies of remdesivir as part of its testing the drug in Ebola, a stockpile that it is now using to provision the studies in China.
But should those tests or trials in the U.S. and elsewhere prove positive, Gilead will need to manufacture enormous quantities of the drug to meet what would likely be extremely high demand from governments and patients.
Gilead has said it is expanding its network of manufacturing partners and has started internal production of remdesivir in preparation of getting coronavirus data.
"We're investing heavily in our supply chain and manufacturing such that, should it work, we can respond immediately," said Gilead's O'Day.
"We're investing tens of millions of dollars today, hundreds of millions of dollars in the very near future to scale up manufacturing facilities — both at Gilead and through a variety of partners — to make this happen," he added.
Another pressing question is remdesivir's potential price, if proven and approved for emergency use.
Analysts at RBC Capital Markets expect Gilead would price the drug at around $900 to $1,000 or lower per course, a cost above oral flu drugs but below new intravenous antibiotics.
How Gilead approaches this question could be a potential flash point between it and national governments, although the pressure of the moment would likely factor into any decision Gilead might make.
For now, O'Day says the company hasn't been discussing pricing in its conversations with governments about clinical trials and compassionate use.
"The topic of pricing comes up once you know whether the medicine works," O'Day said.
"It's very difficult to talk about pricing until you know what value this medicine may or may not bring to patients and to societies," he said. "Having said that, we understand the public health need here and we have to be very conscious about, should this work, how do we get this in the hands of as many patients around the globe as fast as we can."