BioPharma Dive, as part of Emerging Biotech Weekly, is taking a closer look at competitive areas of startup activity. We aim to give an overview of the companies developing a new technology and what their goals are. This State of Play, on GPCR-targeting drugs, is our eighth.
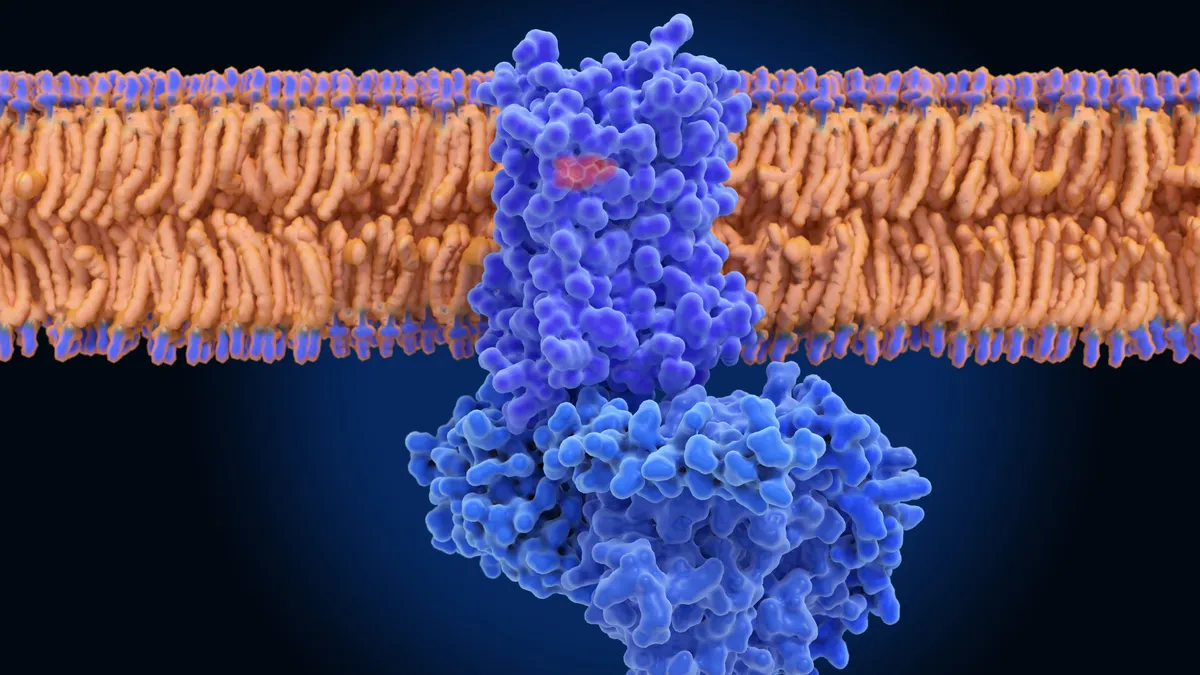
Stationed on the perimeter of cells, the proteins are akin to a neighborhood watchdog. Called G protein-coupled receptors, or GPCRs for short, they handle many of the signals that pass through the cell’s membrane.
Unsurprisingly given their important role, they’re one of the top targets in the drugmaking playbook. According to one estimate, a third of medicines approved by the Food and Drug Administration through 2017 targeted a GPCR in some fashion.
GPCRs were first uncovered in the 1970s by Robert Lefkowitz, a Duke University and Howard Hughes Medical Institute researcher. At the time, GPCRs were a mystery to scientists, who had no way to see their structures.
A series of breakthroughs culminated in the early 2010s, when Lefkowitz and Stanford University scientist Brian Kobilka published research on the structure and function of GPCRs, winning the 2012 Nobel Prize in Chemistry for their efforts.
Their work provided “tremendously detailed” information about how those proteins behave, said Kenneth Jacobson, a senior investigator at the National Institute of Diabetes and Digestive and Kidney Diseases. “As a chemist, that's extremely helpful for searching for new compounds” that can impact GPCRs.
Scientists also now know the receptors can signal via multiple pathways, Jacobson added. That means researchers can hunt for more selective molecules, potentially avoiding off-target effects.
Young biotechnology companies have sensed an opportunity, which is why several startups have formed in recent years hoping to develop better GPCR drugs. Here's where they stand.
What are GPCR-targeting drugs, and how do they work?
GPCR-targeting drugs have a broad variety of potential applications, from possible uses treating cancer to managing diabetes and obesity. In cancer, for instance, the receptors can control tumor growth and spread through activating certain cell functions.
Currently available drugs aimed at GPCRs include antihistamines and opioid agonists. Other examples include the diabetes drug Victoza and the multiple sclerosis treatment Gilenya.
While hundreds of GPCR-targeting medicines are approved, they generally are aimed at a small number of specific GPCRs — leaving room for scientists or drugmakers who want to try elsewhere.
“Most of the success has been around a relatively small number of these GPCRs,” said Jeff Finer, the CEO of Septerna, a startup co-founded by Lefkowitz that’s developing drugs to target the proteins. “There's hundreds more that could be potential therapeutic opportunities.”
For years, though, researchers have struggled to isolate GPCRs, making it harder to design drugs. The methods used to remove the proteins from their surrounding cellular membranes can also inadvertently destroy them. GPCRs are not made in very large quantities by the body, either, upping the challenge.
As a result, pharmaceutical companies researching how to drug the receptors frequently came up short, challenged by both mapping the proteins and by side effects that emerged during clinical trials.
Now, biotechs are trying to develop technologies that allow them to squeeze GPCRs out of their cells more easily, so they can identify possible drug compounds that can attach themselves to the receptors.
What are startups developing GPCR drugs doing differently now?
Drugmakers’ past challenges going after GPCRs has left opportunities for others. Newly formed startups aim to explore areas that were previously ignored or bypassed because they were considered harder to reach.
“An undruggable target could include orphan receptors, olfactory receptors or receptors that might have drugs, but the drugs have too many side effects,” Jacobson said.
Some GPCRs, meanwhile, aren’t as easily reached with small molecules, historically researchers’ go-to tool. Those tough targets often have large "ligands," or signaling molecules, according to Alise Reicin, CEO of Tectonic Therapeutic, a company developing biologic drugs against GPCR targets. “You don’t get enough real estate with a small molecule,” she said.
And biotechs are going after different parts of the receptors, too, buoyed by advancements in protein engineering. Many existing GPCR-targeting drugs look for where ligands bind to a receptor's active site. Developers of the next generation of these drugs are eyeing other nooks and crannies to bind to the protein.
Structure-guided drug discovery, meanwhile, could further optimize the types of medicines developed.
Which companies are developing GPCRs?
Earlier this year, Structure Therapeutics went public with the ticker symbol ‘GPCR,’ advertising its focus in a sign readily recognizable to investors on Wall Street. The $161 million it raised represented one of the largest initial public offerings in biotech over the past twelve months.
Structure is not the only emerging biotech laying claim to new frontiers in the GPCR space, though. Septerna, funded by major biotech investors such as Third Rock Ventures and Samsara BioCapital, launched last year with a $100 million Series A round.
Escient Pharmaceuticals is among the most richly funded, with around $238 million raised in private financing since its launch in 2018.
Structure, Escient and Septerna are joined by nearly half a dozen others that have launched in recent years, raising roughly $900 million combined.
A select list of startups working on new GPCR-targeting drugs
| Company | Top investors | Funding |
|---|---|---|
| Structure Therapeutics | BVF Partners, F-Prime Capital | $359 million |
| Escient Pharmaceuticals | 5AM Ventures, Sanofi Ventures, New Enterprise Associates | $238 million |
| Septerna | Third Rock Ventures, Samsara BioCapital | $100 million |
| Tectonic Therapeutics | Vida Ventures, T.A. Springer, and Polaris Partners | $80 million |
| Confo Therapeutics | BGV, Wellington Partners | “over €40 million” |
| Domain Therapeutics | Panacea Venture, CTI Life Sciences Fund and 3B Future Health Fund | $42 million |
| Teon Therapeutics | NLVC, Oceanpine Capital | $37 million |
| Maxion Therapeutics | Life Arc Ventures | $16 million |
SOURCE: Companies
What is the status of these newer drugs?
Structure has two GPCR drugs in Phase 1 clinical trials: one for diabetes and obesity, the other for a couple of disorders affecting the lung and heart.
Escient has put one of its drug candidates, dubbed EP547, into early-stage clinical testing in conditions associated with end-stage kidney disease and liver diseases.
Pharma companies are buying into others, injecting cash into drug development plans. Last week, Belgium-based Confo Therapeutics announced a licensing deal with Eli Lilly, securing $40 million upfront in exchange for Lilly gaining rights to CFTX-1554, a non-opioid painkiller in a Phase 1 trial.
Merck and Co. is collaborating with Teon Therapeutics, while Lilly and Japanese pharma Sosei Heptares announced a deal in December to develop GPCR-targeting drugs.
AbbVie did the same with Sosei in August and also acquired a GPCR drug startup, DJS Antibodies, in October. Johnson & Johnson is developing a GPCR drug candidate for multiple myeloma.