Sarepta Therapeutics on Monday added to a small, but growing, body of evidence that one of its experimental gene therapies might help treat a potentially deadly genetic disease called limb-girdle muscular dystrophy.
The results aren't yet conclusive. They come from six patients in an early-stage study Sarepta is running to identify the best dose for further testing. It's unclear whether the treatment, known as SRPT-9003, will reverse the course of the disease, and if so, how long the effects will last.
But the data nonetheless show all six are producing what look to be meaningful amounts of a key protein, and the benefit is more significant on a higher dose with which Sarepta is likely to move forward. Additionally, the three who have been followed for a least a year have experienced functional improvements when historical data suggests they shouldn't. Each is able to stand up, walk, run and climb stairs faster than they were able to at the study's start — a notable benefit in a disease that's characterized by progressive muscle weakening.
So far, Sarepta has observed just two serious side effects: a previously disclosed increase in liver enzymes in one of the original three patients treated, and a bout of nausea and vomiting that required hospitalization in one of the more recently treated patients. Both recovered within a few days.
The results are meaningful for multiple reasons, said Sarepta CEO Doug Ingram. Sarepta is developing six different gene therapy programs for various genetic subtypes of limb-girdle. Each of those treatments — as well as a closely watched Duchenne muscular dystrophy program in late-stage testing — have some of the same components as SRP-9003, among them the viral vector used to deliver the gene therapy. Sarepta is using the similarly high dose of that vector in testing of its limb-girdle and Duchenne treatments.
The results thus "adds additional confidence around" all of those programs, Ingram said.
Sarepta estimates between 200,000 to 300,000 people worldwide have limb-girdle muscular dystrophy, which causes a progressive loss of muscle function and often an early death from heart or lung problems. Sarepta's six gene therapies are meant to treat a majority of those with Type 2, which covers some 80% to 90% of cases. SRP-9003 is for people with Type 2E.
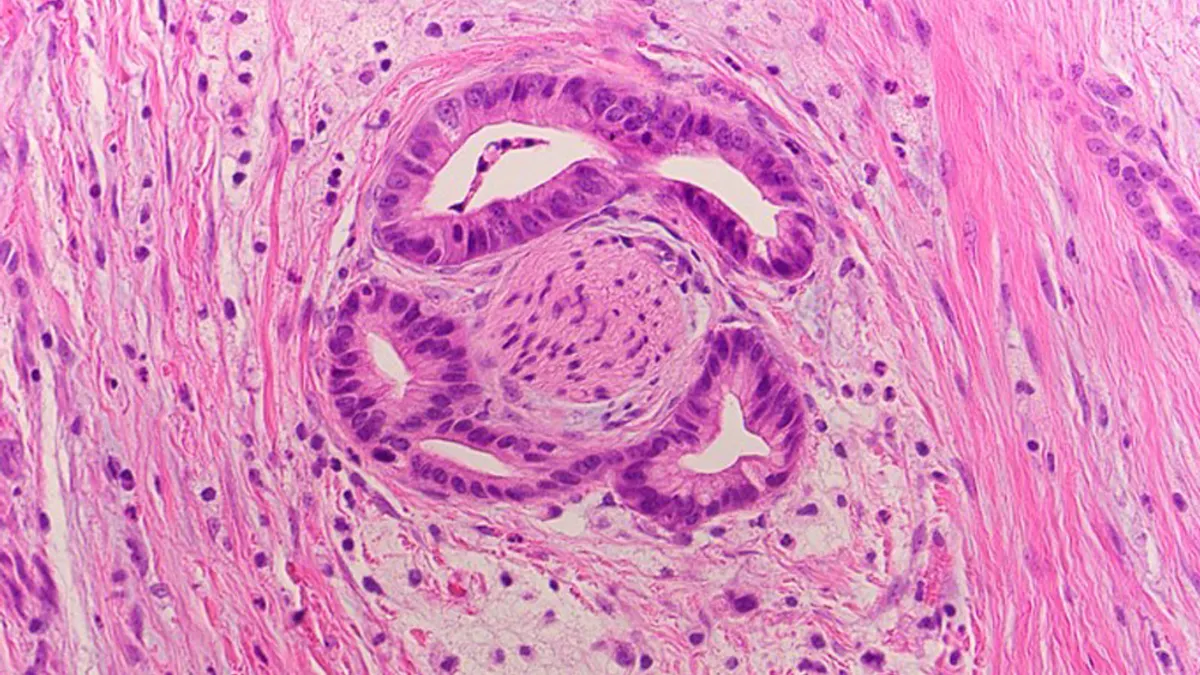
The treatments instruct the body to produce a protein that these patients lack — in SRP-9003's case, beta sarcoglycan. According to Ingram, getting patients to express 20% of normal levels of the protein should lead to a "functional benefit" for patients and change the course of their disease.
Last year, Sarepta reported that two months after treatment, the three patients on a low dose of SRP-9003 had an average of 51% of muscle fibers expressing beta SG and were producing protein levels that were, on average, 36% of normal.
Sarepta now reports even better results for the three patients on a high dose: an average of 72% of muscle fibers with beta SG and a mean of 62% of normal protein expression two months after treatment. Both low and high dose patients have had roughly 90% decreases in levels of creatine kinase, a biological marker of muscle damage.
Clinical improvement in the three low-dose patients resulted in positive changes on a scale known as the North Star Assessment scale that measures motor function in patients with limb-girdle. Sarepta previously disclosed results after nine months of follow-up, but now reports that those changes have held up, and even improved further, after a year. It doesn't yet have this type of data for patients treated with the high dose.
The one new serious side effect Sarepta reported, a patient who was dehydrated from vomiting, resolved after the patient received intravenous fluids and anti-nausea medications. Ingram said the patient had a preexisting gastrointestinal issue that could have been a factor. Other side effects have been mild or moderate.
Since changing its study protocol to extend use of steroids, Sarepta hasn't seen the spike in liver enzymes that it had observed previously — a common gene therapy side effect.
"If it was intolerable, we'd wonder where to go," Ingram said of the higher dose. "But the great news is the safety profile is similar to the first cohort" on the lower dose.
Sarepta will likely use the higher dose, then, for a registrational study that it plans to design with the Food and Drug Administration's help. It's unclear what that will look like, because the only medications available for limb-girdle ease symptoms, rather than change how the disease progresses. But those discussions will help dictate how Sarepta develops its other limb-girdle gene therapies, according to Ingram. Two of them are already in human studies.
Ingram expects Sarepta to be in a position to communicate its plans by early next year.
Sarepta, over the past few years, has evolved from an RNA drug developer to become one of the leaders in the field of gene therapy. The company has done so, in part, by closely aligning itself with Nationwide Children's Hospital in Ohio, through which it has acquired 10 gene therapy programs. Sarepta got five limb-girdle programs when it bought Nationwide spinout Myonexus in February 2019, and licensed the sixth from a Nationwide investigator a few months later. Its pivotal studies in Duchenne should produce results next year.
Ned Pagliarulo contributed reporting.