Biotech: Page 85
-
 National Institutes of Allergy and Infectious Diseases. (2016). "Human natural killer cell" [Micrograph]. Retrieved from Flickr.
National Institutes of Allergy and Infectious Diseases. (2016). "Human natural killer cell" [Micrograph]. Retrieved from Flickr.
Beigene expands into cancer cell therapy with Shoreline pact
The fast-growing biotech will work with Shoreline to develop cancer treatments using natural killer cells, an emerging alternative to T-cell based therapies.
By Ned Pagliarulo • June 9, 2021 -
Deep Dive // New Alzheimer's drugs
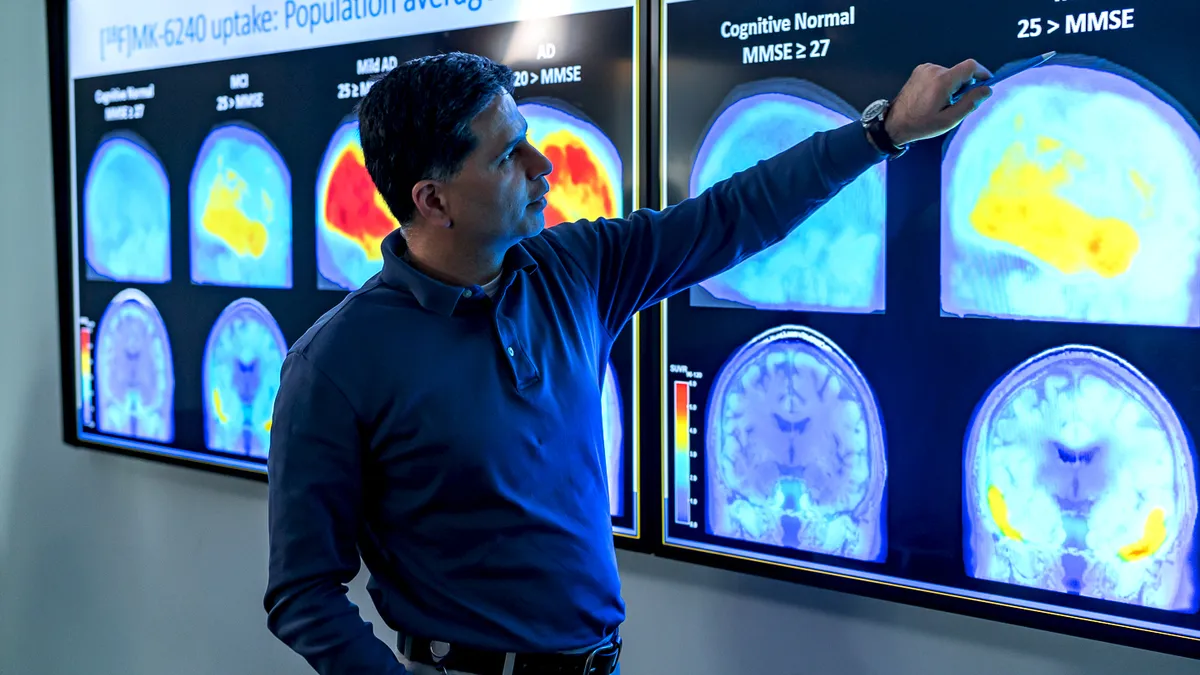
A first-of-its-kind Alzheimer's drug raises heavy questions around who will and won't get it
Biogen priced its newly approved medicine Aduhelm at an average cost of $56,000 a year, adding affordability to other barriers patients may face.
By Jacob Bell • June 8, 2021 -
 Explore the Trendline➔
Explore the Trendline➔
 Spencer Platt via Getty Images
Spencer Platt via Getty Images Trendline
TrendlineEmerging biotech
New biotechs continue to emerge despite a challenging market environment that has forced venture firms to build their drug startups more cautiously.
By BioPharma Dive staff -
The FDA approved Biogen's Alzheimer's drug. The company now has years to confirm it works.
Officially, Biogen has until 2029 to complete a study confirming Aduhelm's benefit. But the agency says it hopes to push the drugmaker to finish faster.
By Ned Pagliarulo • Updated June 9, 2021 -
Deep Dive // New Alzheimer's drugs
In historic move, FDA approves a closely watched and controversial Alzheimer's drug
The decision cleared the way for what many predicted would become a lucrative treatment. But a high price and controversy over whether Biogen's Aduhelm benefits patients weighed heavily on the drug's launch.
By Jacob Bell • Updated June 7, 2021 -
Bluebird cleared by FDA to resume studies of sickle cell gene therapy
An investigation by the company determined the treatment was "very unlikely" to be the cause of a case of leukemia reported in one of the trials.
By Ned Pagliarulo • June 7, 2021 -
Orchard abandons promising gene therapy for rare immune disorder
The U.K. biotech deprioritized the therapy last year, despite positive results in clinical testing that were recently published in a top medical journal.
By Kristin Jensen • June 3, 2021 -
Biotech venture capital still booming as venBio raises another $550M
VenBio has previously invested in a range of treatment areas and technologies, including cell and gene editing companies like Precision Bio and Artiva.
By Jacob Bell • June 3, 2021 -
Bayer buys a biotech and its offshoot in bet on radiopharmaceuticals
The German pharma will add an experimental prostate cancer medicine to its pipeline through the deal, which reflects growing industry interest in the field.
By Ned Pagliarulo • June 3, 2021 -
MorphoSys to buy Constellation in $1.7B deal, aided by unusual funding
Royalty Pharma will effectively fund MorphoSys' acquisition of the cancer biotech through a separate agreement to buy MorphoSys' shares of royalties on several marketed and experimental medicines.
By Ned Pagliarulo • June 2, 2021 -
Alkermes prepares for uncertain launch as FDA finally clears schizophrenia drug
Lybalvi is meant to be as effective as marketed antipsychotics but without the weight gain patients typically experience. Alkermes will introduce the drug into a competitive market, however.
By Jacob Bell • June 1, 2021 -
Amgen pays $400M for an eczema drug, putting faith in an old partner
A deal with Kyowa Kirin gives Amgen rights to an experimental treatment aimed at an autoimmune target that has drawn interest from multiple drugmakers.
By Kristin Jensen • June 1, 2021 -
Sponsored by Parexel Biotech
4 insights on decentralized trials for biotechs
Here are four things we have learned about DCTs that biotech companies can consider when designing a decentralized or hybrid trial.
June 1, 2021 -

 Retrieved from National Cancer Institute on September 27, 2019
Retrieved from National Cancer Institute on September 27, 2019
In first, FDA approves KRAS-blocking cancer drug from Amgen
For decades, scientists have tried unsuccessfully to target the KRAS gene, which is often mutated in lung, colon and pancreatic cancers. Lumakras is the first drug proven effective.
By Ned Pagliarulo • Updated May 29, 2021 -
EQRx readies a lower-cost alternative to pricey cancer immunotherapies
The biotech and its partner CStone disclosed Phase 3 results in lung cancer that support approval plans for a drug similar to treatments like Merck's Keytruda, but would be priced at a fraction of the cost.
By Ben Fidler • May 28, 2021 -
Centessa raises $330M in third largest biotech IPO this year
The public offering follows just three months after Centessa, created from the merger of 10 Medicxi-backed drug startups, launched with $250 million in venture funding.
By Ned Pagliarulo • May 28, 2021 -
UniQure moves Huntington's gene therapy to next phase of key trial
Safety monitors cleared the company to test a higher dose after reviewing data from 10 patients, providing some hopeful news after recent setbacks for experimental drugs targeting the genetic disease.
By Jonathan Gardner • May 27, 2021 -
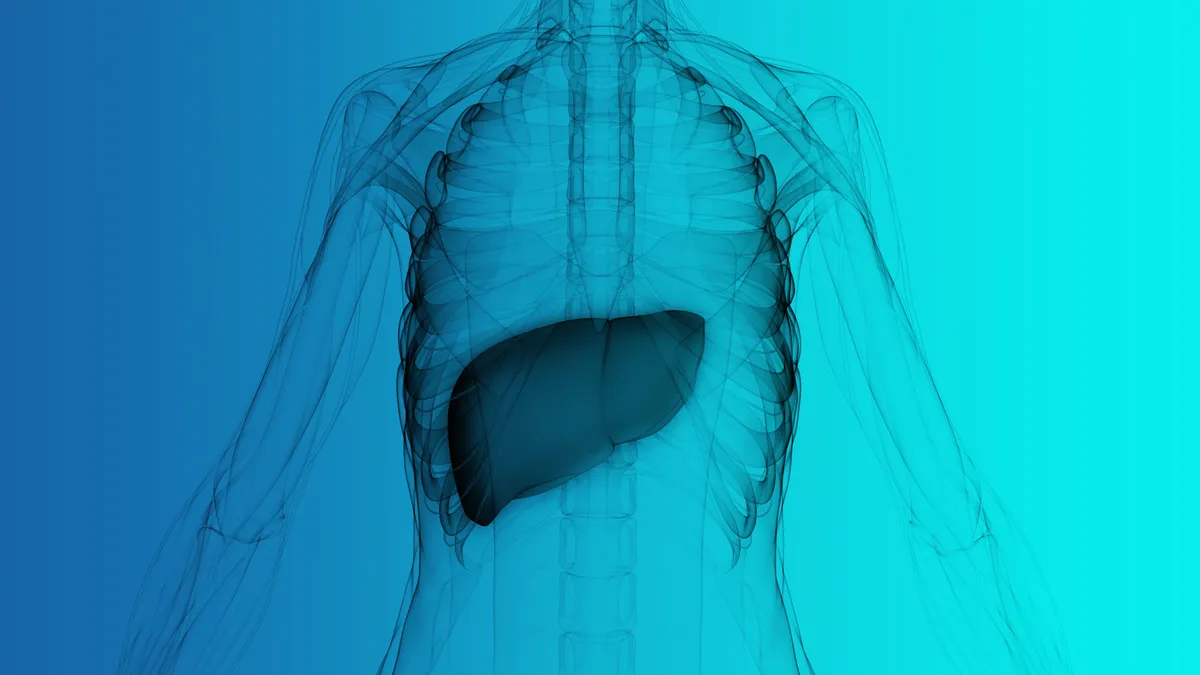
FDA restricts use of Intercept drug due to liver injury risk
An investigation linked the drug to severe injuries in about two dozen patients, leading the agency to add a new contraindication to the label.
By Kristin Jensen • May 27, 2021 -
'A hugely consequential decision:' How Biogen's Alzheimer's drug came to face the FDA
The FDA's decision to approve aducanumb could have far-reaching consequences for patients, Biogen and Alzheimer's research. Here's how the drug's review came about.
By Ned Pagliarulo • May 27, 2021 -
Lyell aims to follow Juno, Sana as Wall Street cools to biotech IPOs
The ambitious cell therapy developer, which has already raised nearly $1 billion from investors, will test the IPO market just as the pace and performance of recent biotech offerings have slowed considerably.
By Ben Fidler • May 26, 2021 -
J&J's new cancer drug leads a growing pipeline of dual-targeting antibodies
Pharma and biotech companies alike see potential in using bispecific antibodies for hard-to-treat cancers like leukemia, myeloma and solid tumors.
By Jonathan Gardner • May 24, 2021 -
A biotech backs off NASH after trial failure
NGM Biopharmaceuticals has decided not to pursue a late-stage NASH program after its drug aldafermin became the latest in a long line of medicines to fail to help patients with the common liver condition.
By Jacob Bell • May 24, 2021 -
Bluebird's next gene therapy gets backing from European regulator
The treatment for a progressive, often deadly brain disease could soon become Bluebird's third approved product and one of only a handful of marketed gene therapies in the world.
By Jacob Bell • May 21, 2021 -
In the midst of a 'strategic shift,' Voyager loses CEO and R&D head
The shake-ups come as the biotech prepares to invest more in its technology and also begin human testing for one of its most advanced gene therapies.
By Jacob Bell • May 20, 2021 -
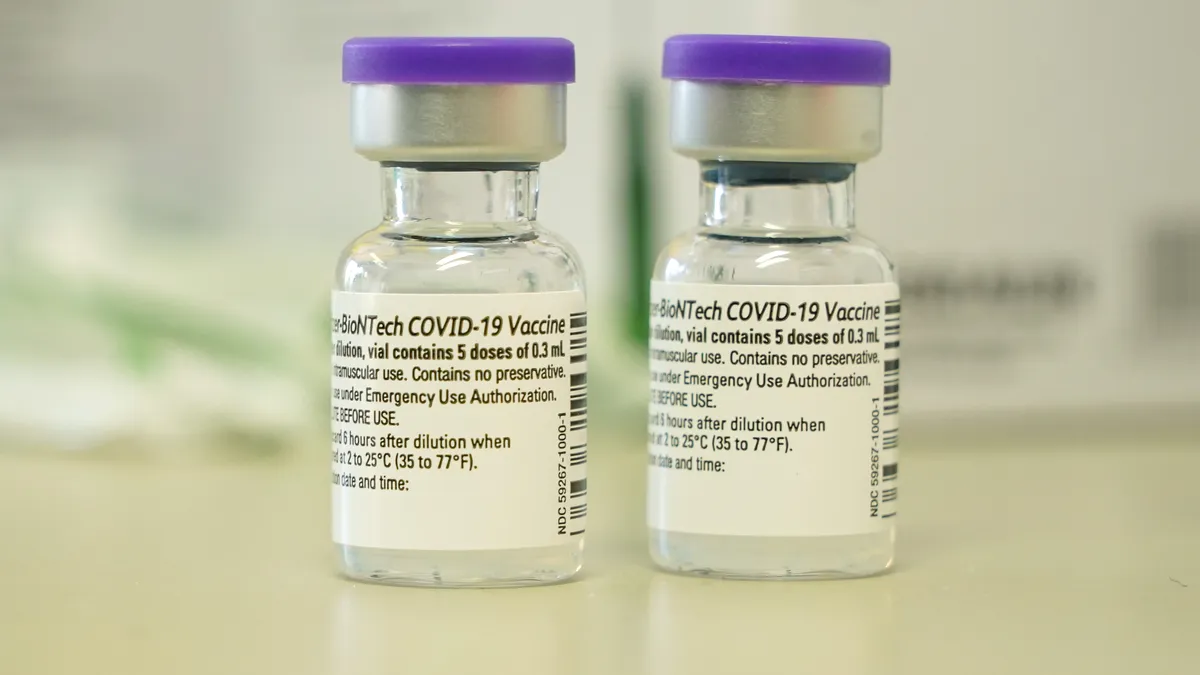
Pfizer, BioNTech to send EU up to 1.8B coronavirus vaccine doses
The deal, which the companies and the bloc have been negotiating for weeks, will significantly expand on the current contract for 600 million shots.
By Kristin Jensen • May 20, 2021 -
Off-the-shelf CAR-T, a Tagrisso rival and the next breast cancer drugs
Study abstracts for drugs from Allogene, EQRx, Sanofi and Lilly drew early interest and helped set the stage for the meeting.
By Ben Fidler , Ned Pagliarulo • May 20, 2021