Marketing: Page 5
-
Walgreens to be acquired in $10B take-private deal
The struggling retail pharmacy chain entered an agreement to be purchased by private equity firm Sycamore Partners, which could end its nearly 100-year run as a public company.
By Emily Olsen • March 7, 2025 -
Pain drugs
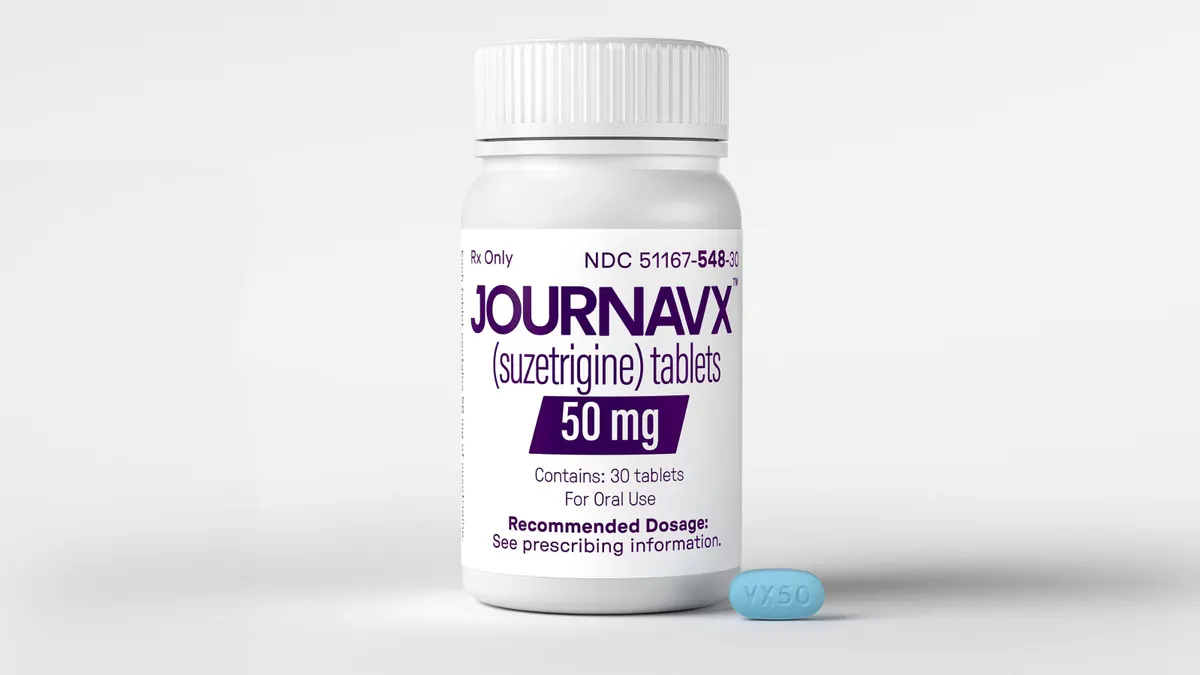
Vertex’s new pain drug gets first coverage nod from major insurer
Optum Rx, the pharmacy benefit manager owned by UnitedHealth Group, placed Journavx on some of its commercial formularies in an interim decision while a formal review continues.
By Jacob Bell • March 6, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Brian Tucker / BioPharma Dive/BioPharma Dive
Brian Tucker / BioPharma Dive/BioPharma Dive Trendline
TrendlineCommercialization
New drugs for obesity are becoming blockbusters, while Trump administration pressure is reshaping pharma marketing strategies ahead of looming patent cliffs.
By BioPharma Dive staff -
Leqembi, after delay, gets back on track toward EU approval
European regulators “reaffirmed” a positive view of Eisai and Biogen’s Alzheimer’s drug after conducting a new safety review.
By Jonathan Gardner • Feb. 28, 2025 -
Obesity drugs
Drug compounders sue FDA over declaration ending Wegovy shortage
Telehealth provider Hims & Hers said it will tell consumers they need to seek “alternative options on the commercial dosing,” but in earnings Monday still forecast higher revenue and profits in 2025.
By Jonathan Gardner • Feb. 25, 2025 -
FTC case against PBMs can move forward, judge rules
A Missouri district court judge said stopping the FTC’s suit against Caremark, Express Scripts and Optum Rx would be “against the public’s interest.”
By Rebecca Pifer Parduhn • Feb. 20, 2025 -
BridgeBio’s heart drug launch gets off to a fast start
Prescription totals for Attruby, which is locked in a closely watched commercial battle with Pfizer’s tafamidis, have more than doubled since January and outpaced investor expectations.
By Ben Fidler • Feb. 20, 2025 -
Axsome secures top drug’s future with Teva patent settlement
Shares in the brain drug developer rose by more than 20% Monday on news of the deal, which will forestall Teva's generic copy of Auvelity from entering the U.S. market until at least 2038.
By Ned Pagliarulo • Feb. 10, 2025 -
Brain drug revival
Bristol Myers gives first peek at closely watched launch of schizophrenia drug
Cobenfy, the main asset acquired through Bristol Myers’ $14 billion purchase of Karuna, had $10 million in sales in the fourth quarter and around 1,000 prescriptions weekly by late January.
By Jacob Bell • Feb. 6, 2025 -
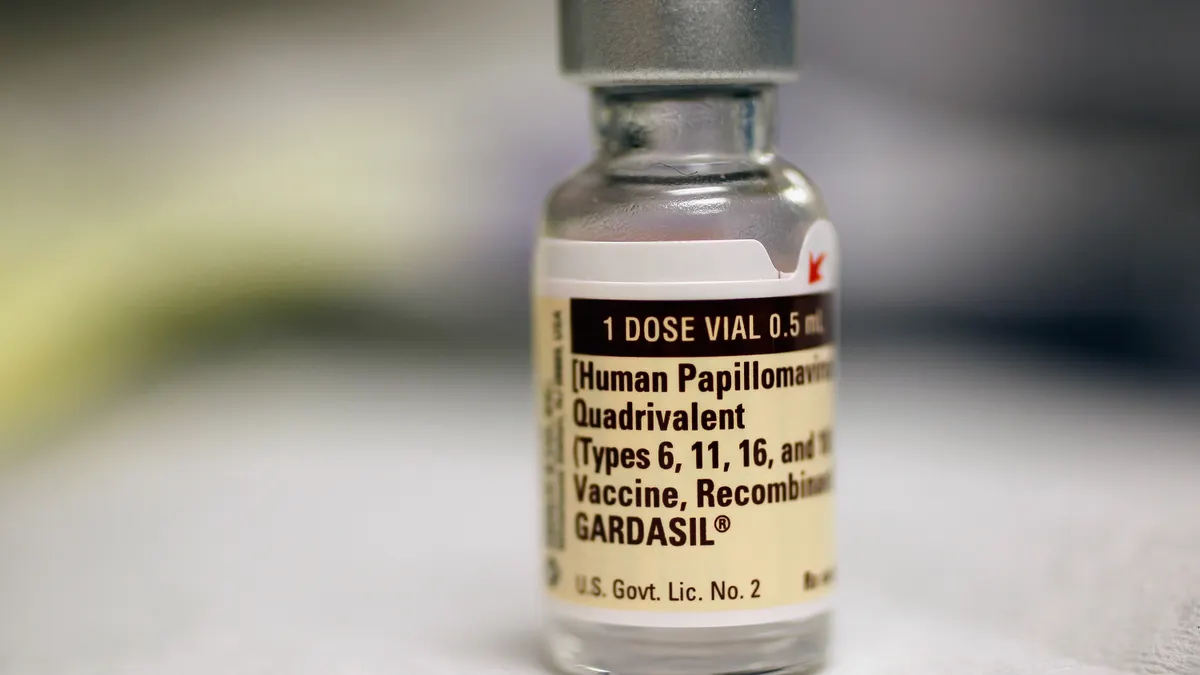
Merck shares tumble as company pauses Gardasil shipments to China
Executives pulled back on a 2030 sales target of $11 billion as demand for the HPV shot shrinks amid China market turmoil.
By Jonathan Gardner • Feb. 4, 2025 -
Judge halts Trump freeze on grants as states report healthcare payment portals back online
States and providers regained access to a key healthcare payments portal late Tuesday amid pushback over the Trump administration’s decision to halt federal assistance.
By Rebecca Pifer Parduhn • Jan. 29, 2025 -
Q&A
BridgeBio’s Neil Kumar on an underdog drug launch and wooing deal-hungry investors
In an interview, the CEO discussed trying to gain support from investors hyper-focused on M&A, and competing with Pfizer and Alnylam.
By Ben Fidler • Jan. 23, 2025 -
Sponsored by Scientist.com
Navigating future biopharma catalysts: What to expect at the 2025 J.P. Morgan Healthcare Conference
The stage is set for JPM 2025—don't miss expert reactions following 2025’s principal catalyst event!
Jan. 6, 2025 -
GLP-1 drug compounding is in limbo. Will the FDA draw out its decision?
The agency is due to make a determination on the shortage status of Zepbound by Dec. 19. But another delay could be possible and, either way, experts predict more litigation.
By Amy Baxter • Dec. 11, 2024 -
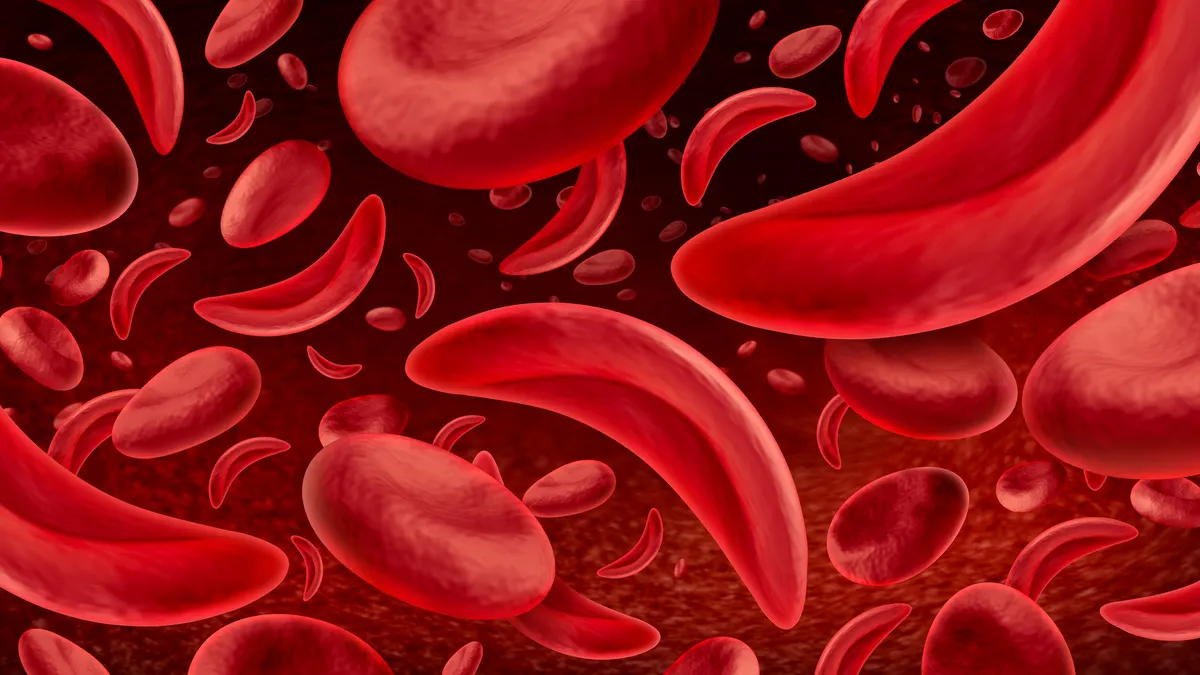
Gene therapy uptake in sickle cell stays slow, despite patient interest
A lengthy treatment process, coupled with weighty risks for recipients to consider, has resulted in plodding adoption of Casgevy and Lyfgenia during their first year on market.
By Ned Pagliarulo , Gwendolyn Wu • Dec. 9, 2024 -
Sponsored by Almac Clinical Services
Clinical services organizations are critical to the future of pharma in the APAC market
As APAC contract manufacturing grows, pharma companies turn to outsourcing for cost-effective solutions.
Dec. 9, 2024 -
Diabetes advocacy group discourages use of compounded GLP-1 drugs
The ADA recommended doctors avoid prescribing unapproved, off-brand versions of drugs like Wegovy, wading into a dispute that has gripped the FDA and compounding pharmacies over the past few months.
By Jonathan Gardner • Dec. 2, 2024 -
Biden administration proposes Medicare, Medicaid coverage of pricey weight loss drugs
Millions of Americans who have obesity could gain insurance coverage for drugs like Wegovy if the Trump administration allows the rule to go into effect.
By Rebecca Pifer Parduhn • Nov. 26, 2024 -
Will Walgreens’ store closures disrupt its clinical trial aims?
Despite plans to shutter 1,200 stores, the company’s chief clinical trials officer said the pharmacy giant remains committed to aiding drug research.
By Meagan Parrish • Nov. 22, 2024 -
Under-the-skin Keytruda comparable to infused version in Phase 3 study, Merck says
Merck plans to discuss the data with regulators as it lags rivals Roche and Bristol Myers in bringing forward a subcutaneous form of its cancer drug.
By Jonathan Gardner • Nov. 19, 2024 -
Eli Lilly follows J&J in suing Biden administration over 340B rebates
Major drugmakers are filing lawsuits against the HHS and HRSA to resuscitate plans to issue hospitals rebates instead of giving them upfront discounts on 340B drugs.
By Rebecca Pifer Parduhn • Nov. 18, 2024 -
New Alzheimer's drugs
Eisai wins over European regulators on Alzheimer’s drug Leqembi
The EMA sided against the drug this summer. But an appeal from Eisai appears to have worked, teeing Leqembi up for authorization in a major market.
By Jacob Bell • Nov. 14, 2024 -
J&J sues US government for halting 340B rebate plan
The pharma is trying to win court backing to pay hospitals rebates for two drugs in the 340B drug discount program, instead of offering upfront discounts.
By Rebecca Pifer Parduhn • Nov. 13, 2024 -
Cigna confirms it is not pursuing Humana acquisition
The formal denial comes after Cigna CEO David Cordani tried to push back on persistent speculation of a Humana merger earlier this fall.
By Rebecca Pifer Parduhn • Nov. 11, 2024 -
Sponsored by SmartLabs
Thinking inside the box: How SmartLabs is driving new collaborations to support the life science industry in global innovation hubs
Seamlessly scale and adapt to match the rapid pace and complexity of modern science.
Nov. 11, 2024 -
Pfizer’s RSV vaccine cleared by FDA for use in some younger adults
Approval of Pfizer's Abrysvo in certain adults aged 18 to 59 years old will expand the number of people eligible for vaccination with the shot.
By Delilah Alvarado • Oct. 23, 2024