Biotech: Page 82
-
After talks with FDA, a biotech prepares to submit its closely watched ALS drug
Executives at Amylyx Pharmaceuticals said that based on the agency's recent feedback, they will soon ask for approval of AMX0035 — a change of plans from earlier this year.
By Jacob Bell • Sept. 15, 2021 -
CureVac scales back mRNA manufacturing for coronavirus vaccine
The German biotech, which announced disappointing results for its shot in June, said the move was done in response to reduced demand.
By Shoshana Dubnow • Updated Sept. 15, 2021 -
 Explore the Trendline➔
Explore the Trendline➔
 Spencer Platt via Getty Images
Spencer Platt via Getty Images Trendline
TrendlineEmerging biotech
New biotechs continue to emerge despite a challenging market environment that has forced venture firms to build their drug startups more cautiously.
By BioPharma Dive staff -
Biotech funder Lightstone raises another $375M for life sciences investing
The venture firm, which previously invested in LocanaBio, Catamaran Bio and Tizona Therapeutics, said its third fund surpassed its target.
By Jacob Bell • Sept. 14, 2021 -
Fourth trial volunteer dies in Astellas gene therapy study
The study, which was suspended following three deaths last year, had been restarted in February after Astellas lowered the treatment dose used.
By Ned Pagliarulo • Sept. 14, 2021 -
Biogen offered a window into what's holding up Aduhelm. Investors didn't like what they saw.
At Morgan Stanley’s healthcare conference last week, executives detailed how the launch of their Alzheimer’s drug has been slower than expected, due in large part to lingering questions about trial data and insurance coverage.
By Jacob Bell • Sept. 9, 2021 -
Apellis eye drug succeeds in one key study, but falls short in its twin
Highly anticipated study results showed divergent outcomes for the biotech's therapy in treating a form of vision loss known as geographic atrophy. Even so, Apellis plans to soon ask for FDA approval.
By Ned Pagliarulo • Updated Sept. 10, 2021 -
Jennifer Doudna-founded CRISPR biotech raises new cash to broaden drug development work
Originally formed as a diagnostics developer by Doudna and three other cofounders, Mammoth Biosciences has accelerated plans to make gene editing medicines.
By Shoshana Dubnow • Updated Sept. 10, 2021 -
Moderna expands mRNA vaccine supply in deal with new biotech producer
The company has previously said it could make up to 3 billion vaccine doses in 2022, depending on how booster shots are authorized.
By Shoshana Dubnow • Sept. 8, 2021 -
Venture capital pours more money into RNA medicines with the launch of Replicate
The company, which focuses on self-replicating RNA, comes equipped with $40 million from Apple Tree Partners and a stamp of approval from the former research head at Biogen.
By Jacob Bell • Sept. 8, 2021 -
Roche bets big on off-the-shelf cancer cell therapy with Adaptimmune deal
Long a bystander in the race to develop so-called allogeneic treatments, the cancer drugmaking giant has joined the fray with a wide-ranging alliance that could be worth up to $3 billion.
By Jonathan Gardner • Sept. 7, 2021 -
FDA halts tests of BioMarin drug amid heightened focus on gene therapy safety
On the heels of a two-day FDA meeting in which experts debated the risks of gene therapy, the biotech said its early-stage PKU treatment was associated with cancer in mice.
By Ben Fidler • Sept. 7, 2021 -
 National Institute on Aging. (2017). "Beta-Amyloid Plaques and Tau in the Brain" [Image]. Retrieved from Flickr.
National Institute on Aging. (2017). "Beta-Amyloid Plaques and Tau in the Brain" [Image]. Retrieved from Flickr.
Hint of benefit in Alzheimer's drug study fuel stock surge for Swiss biotech
A once-failed drug being developed by AC Immune and Roche appeared to slow cognitive decline in a small trial. But other study goals were missed, muddying the seemingly positive result.
By Ned Pagliarulo • Aug. 31, 2021 -
Galapagos CEO to retire, though successor yet to be announced
Onno van de Stolpe co-founded Galapagos in 1999. Under his watch, the Belgian biotech secured its first approved drug and a landmark deal with Gilead, though it has since hit a series of setbacks.
By Jacob Bell • Aug. 31, 2021 -
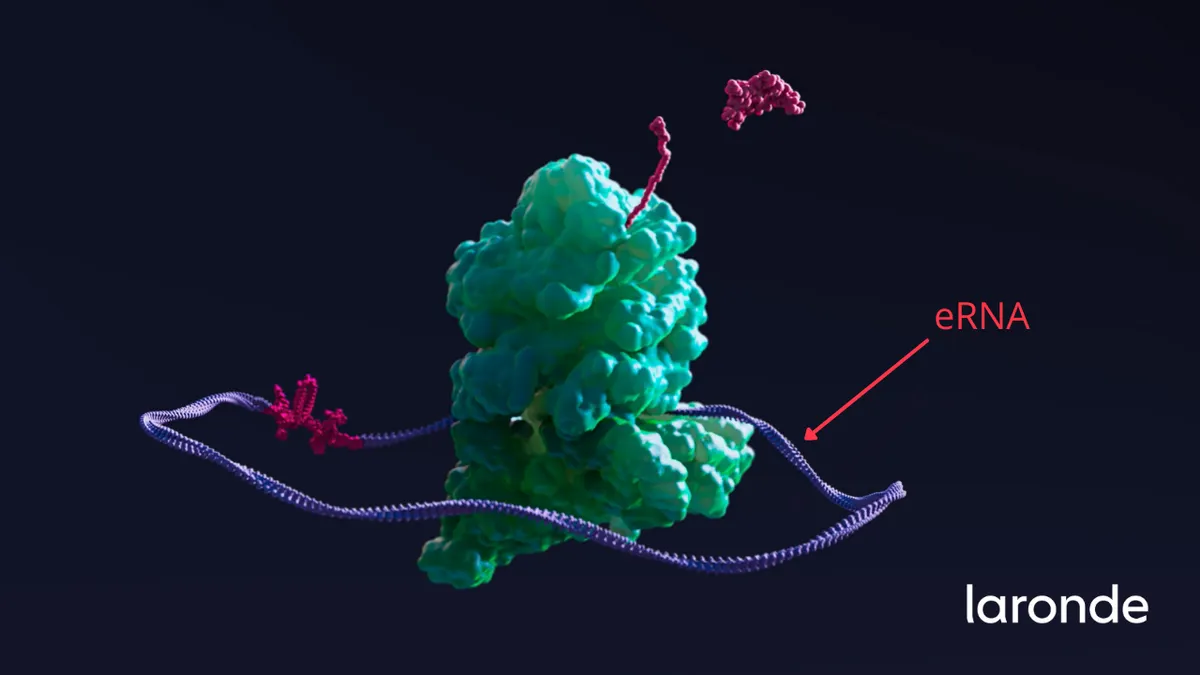
Moderna founder's next big play in RNA raises $440M
Just a few months out of stealth mode, Laronde and its "endless RNA" technology have caught the attention of well-known investors like Fidelity, T. Rowe Price Associates and Invus.
By Jacob Bell • Aug. 30, 2021 -
BioMarin wins European approval to sell drug for dwarfism
The drugmaker is setting the price for Voxzogo high, at an annual price of around $300,000 per patient, although the company expects to make "material discounts" in the coming years.
By Ned Pagliarulo • Aug. 27, 2021 -
Gilead wins reversal of $1.2B penalty in CAR-T patent case
A federal appeals court overturned an earlier verdict that had ruled Gilead's Kite Pharma unit infringed on a patent held by Bristol Myers Squibb.
By Ned Pagliarulo • Updated Aug. 26, 2021 -
New data bolster Regeneron's plan to defend top-selling eye drug
Early results show a higher dose of the company's flagship drug Eylea appears as safe as the marketed version, a step forward in the company's plan to fight off emerging competition in the years ahead.
By Kristin Jensen • Aug. 25, 2021 -
Vertex, looking to broaden its gene editing abilities, asks a young biotech for help
A freshly inked agreement gives Vertex the right to use tools from Arbor Biotechnology to develop "ex vivo" cell therapies for diseases like Type 1 diabetes and sickle cell, among others.
By Jacob Bell • Aug. 24, 2021 -
With new results, J&J's $1B gamble on a targeted inflammation drug faces long odds
Theravance, which agreed to co-develop a next-generation JAK inhibitor with J&J three years ago, said the experimental drug failed a mid-stage study in ulcerative colitis, leaving its future in doubt.
By Jonathan Gardner • Aug. 24, 2021 -
 National Institutes of Allergy and Infectious Diseases. (2016). "Human natural killer cell" [Micrograph]. Retrieved from Flickr.
National Institutes of Allergy and Infectious Diseases. (2016). "Human natural killer cell" [Micrograph]. Retrieved from Flickr.
With new Fate data, same promise, questions surround 'natural killer' cell therapy
Though early results from two of the biotech's experimental lymphoma treatments have shown promise, it’s unclear whether they can match the potency and durability of T cell therapies.
By Ben Fidler • Aug. 20, 2021 -
Coherus, known for biosimilars, nears an immunotherapy battle with big pharma
New lung cancer results could make Coherus the next entrant in an ultra-competitive market dominated by pricey drugs like Merck's Keytruda.
By Ben Fidler • Aug. 19, 2021 -
Private equity dives deeper into biotech with nearly $2B Bain fundraise
Bain Capital's latest life sciences fund, its third, is another example of private equity's growing interest in drug development.
By Jacob Bell • Aug. 17, 2021 -
Merck wins approval for cancer drug acquired in 2019 biotech buyout
The FDA cleared Welireg, which Merck picked up in its $1 billion deal for Peloton Therapeutics, to treat tumors associated with a rare genetic disease.
By Ned Pagliarulo • Aug. 16, 2021 -
VA leaves Aduhelm off coverage list, recommending against controversial Alzheimer's drug
The decision, which does provide for certain exceptions, is another bump in what's become a rocky market rollout for Biogen's medicine.
By Ned Pagliarulo • Updated Aug. 12, 2021 -
FDA rejects FibroGen's kidney drug in major blow to company
While the decision was expected after a negative advisory panel last month, it's a significant setback that could trigger cost-cutting at the biotech, which said it will discuss next steps with partner AstraZeneca.
By Kristin Jensen • Aug. 11, 2021