Gene Therapy: Page 4
-
Gene editing
Intellia filing spurs safety concerns over CRISPR drug
Shares lost a quarter of their value after the company disclosed, in a regulatory filing, signs of liver stress in a study participant given one of its gene editing treatments.
By Kristin Jensen • May 29, 2025 -
Patient dies in trial of Rocket gene therapy for heart condition
The FDA has placed a clinical hold on the study while Rocket works with trial monitors and experts to investigate the cause of the patient’s death.
By Ned Pagliarulo • May 27, 2025 -
 Explore the Trendline➔
Explore the Trendline➔
 Getty Images
Getty Images Trendline
TrendlineGene Therapy
Rapid scientific advances have put the gene therapy field at the forefront of biomedical research. But, as recent setbacks have shown, researchers and drugmakers still face major challenges.
By BioPharma Dive staff -
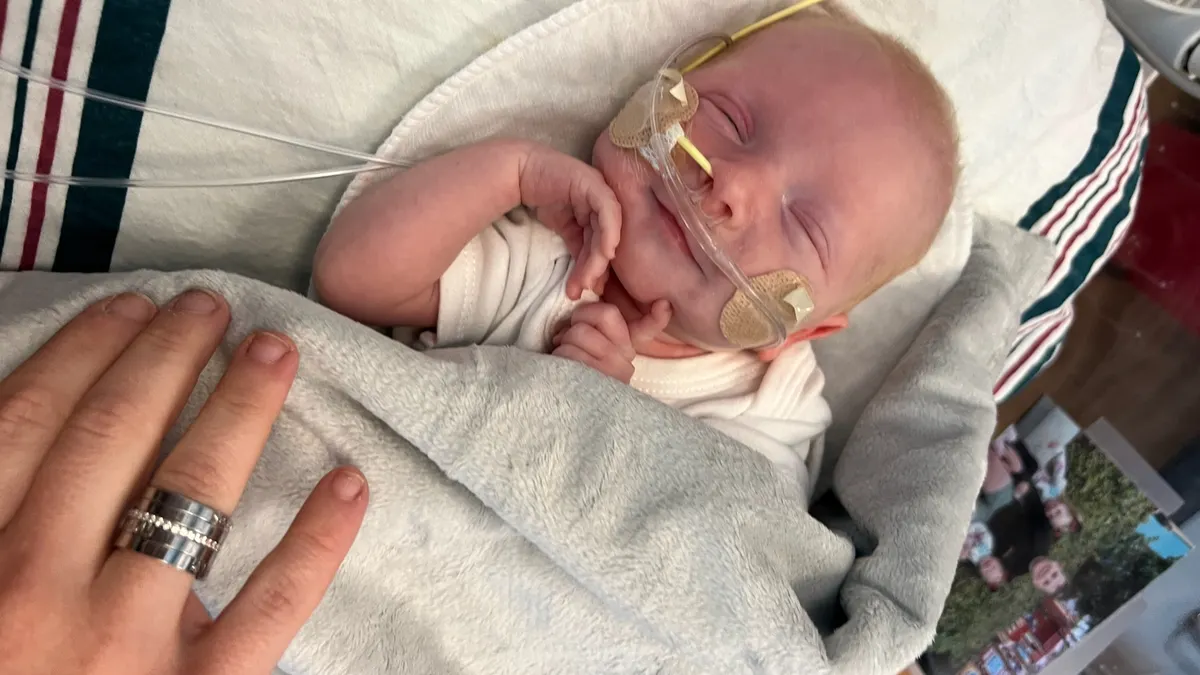
A bespoke CRISPR therapy suggests a blueprint for treating ‘N-of-1’ diseases
A gene editing drug custom-made for a critically ill baby showed that, for some ultra-rare diseases, it’s possible to design and test a new CRISPR medicine in just a few months.
By Ben Fidler • May 15, 2025 -
AbbVie stakes $335M on a startup’s RNAi drugs
ADARx Pharmaceuticals will work with AbbVie to make RNA interference medicines for a variety of disease areas, including neurology, immunology and cancer.
By Gwendolyn Wu • May 15, 2025 -
Abeona sells speedy drug review voucher for $155M
The biotech has quickly turned a cell therapy approval into one of the more lucrative recent sales for a priority review voucher, which companies can use to fast track drug reviews.
By Ben Fidler • May 12, 2025 -
Sarepta shares plummet as lower sales outlook adds to gloom over FDA’s new biologics hire
Already facing headwinds caused by a patient’s death and treatment delays, the biotech now may have to contend with a less friendly FDA.
By Kristin Jensen • May 7, 2025 -
Prasad’s FDA appointment pressures cell and gene therapy stocks
A vocal opponent of his predecessor Peter Marks, Vinay Prasad will now lead the office tasked with reviewing some genetic medicines, adding more uncertainty to an already struggling field of research.
By Ben Fidler • May 6, 2025 -
Vinay Prasad, a physician and FDA critic, to lead agency center overseeing vaccines
Prasad has gained a reputation for questioning U.S. health policy and accelerated approvals in oncology, and has also opposed some vaccine mandates and the use of COVID shots in children.
By Ben Fidler , Jonathan Gardner • May 6, 2025 -
Abeona cell therapy approved by FDA for rare skin condition
Zevaskyn, which Abeona priced at $3.1 million, treats epidermolysis bullosa. It will compete with a gene therapy from Krystal Biotech that is off to a strong sales start.
By Ben Fidler • April 29, 2025 -
 del Aguila III, Ernesto. (2018). "CRISPR Cas9" [Illustration]. Retrieved from Flickr.
del Aguila III, Ernesto. (2018). "CRISPR Cas9" [Illustration]. Retrieved from Flickr. Immune reset
Immune resetCaribou, in reversal, scraps autoimmune cell therapy and cuts staff
The CRISPR company is laying off about a third of its employees and stopping work on an experimental lupus treatment in a bid to focus resources on a pair of cancer cell therapies.
By Ben Fidler • April 25, 2025 -
Bluebird says Ayrmid missed deadline for rival takeover bid
The company’s board reiterated its recommendation to instead pursue an offer from SK Capital and Carlyle that’s worth about $29 million upfront.
By Gwendolyn Wu • April 16, 2025 -
Gene editing
Verve’s second swing at gene editing for heart disease shows early promise
Wall Street analysts said the results for Verve’s therapy appeared competitive on efficacy, while avoiding any major safety concerns — at least so far.
By Ben Fidler • April 14, 2025 -
Sponsored by QIAGEN Digital Insights
The critical role of evidence-based databases in pharmacogenomics: Ensuring accuracy, consistency and relevance
Discover how QIAGEN PGXI transforms pharmacogenomics with faster, more accurate insights!
By QIAGEN Digital Insights • April 14, 2025 -
Roche halts testing of Sarepta Duchenne gene therapy in Europe
The Swiss drugmaker, which holds European rights to Elevidys, suspended three trials while researchers investigate the death of a young man who died following treatment.
By Kristin Jensen • April 3, 2025 -
New genetic medicine CDMO acquires Landmark Bio
Artis BioSolutions joins a host of startups trying to improve development and manufacturing capacity for cutting-edge gene and cell therapies.
By Kristin Jensen • April 2, 2025 -
Cell, gene therapy makers lose a champion at FDA with exit of Peter Marks
Marks’ resignation leaves the field without a regulator many view as “integral” to its progress over the last decade.
By Ben Fidler • March 31, 2025 -
Bluebird gets rival takeout offer from Ayrmid
The new bid surpasses an offer SK Capital and Carlyle Group made last month for the gene therapy developer, which Bluebird’s board still supports.
By Ben Fidler • March 28, 2025 -
Gene editing
Verve gets FDA green light to expand base editing trial into US
The company expects next quarter to read out initial data from its Heart-2 trial, which is testing a one-time treatment for people with genetically elevated cholesterol.
By Gwendolyn Wu • March 24, 2025 -
Novartis builds case for new SMA gene therapy
To Novartis, fresh late-stage data support the idea that its intrathecal drug, which has the same active ingredient as Zolgensma, could be approved for a broad range of spinal muscular atrophy patients.
By Jacob Bell • March 20, 2025 -
Gene editing
Prime sets sights on liver, lung disease as next target for its gene editing tech
The biotech unveiled a new research program in alpha-1 antitrypsin deficiency, a disease that’s also a focus for rival Beam Therapeutics.
By Ned Pagliarulo • March 18, 2025 -
Duchenne patient dies after receiving Sarepta gene therapy
A young man treated with Elevidys died of acute liver failure. The case may give doctors pause before prescribing the treatment in the future, some analysts wrote.
By Ben Fidler • Updated March 18, 2025 -
MeiraGTx spins Parkinson’s, obesity gene therapies into AI startup
The joint venture with generative AI firm Hologen hands MeiraGTx $200 million up front as well as other financial perks — a “transformative” deal, according to the company’s CEO.
By Jacob Bell • March 13, 2025 -
2seventy bio, Bluebird’s cell therapy spinout, sells to Bristol Myers for less than $300M
The deal ends 2Seventy’s short run as an independent company, during which it restructured, sold off research and lost nearly all of its market value.
By Ben Fidler • March 11, 2025 -
Gene editing
Beam base editing therapy gets ‘proof of concept’ in rare lung disease
While initial study results suggest Beam's technology can correct alpha-1 antitrypsin deficiency's genetic roots, shares fell by double digits.
By Ned Pagliarulo • March 10, 2025 -
Sponsored by MilliporeSigma
3 ways to accelerate development and de-risk cell and gene therapy manufacturing
Development strategies designed to reduce the unknowns so that cell and gene therapy innovators can develop robust processes efficiently.
March 10, 2025