Dive Brief:
- European drug regulators have again concluded that an ALS medicine developed by Amylyx Pharmaceuticals should not be cleared for market, citing concerns about the treatment’s overall effectiveness and the reliability of the data supporting it.
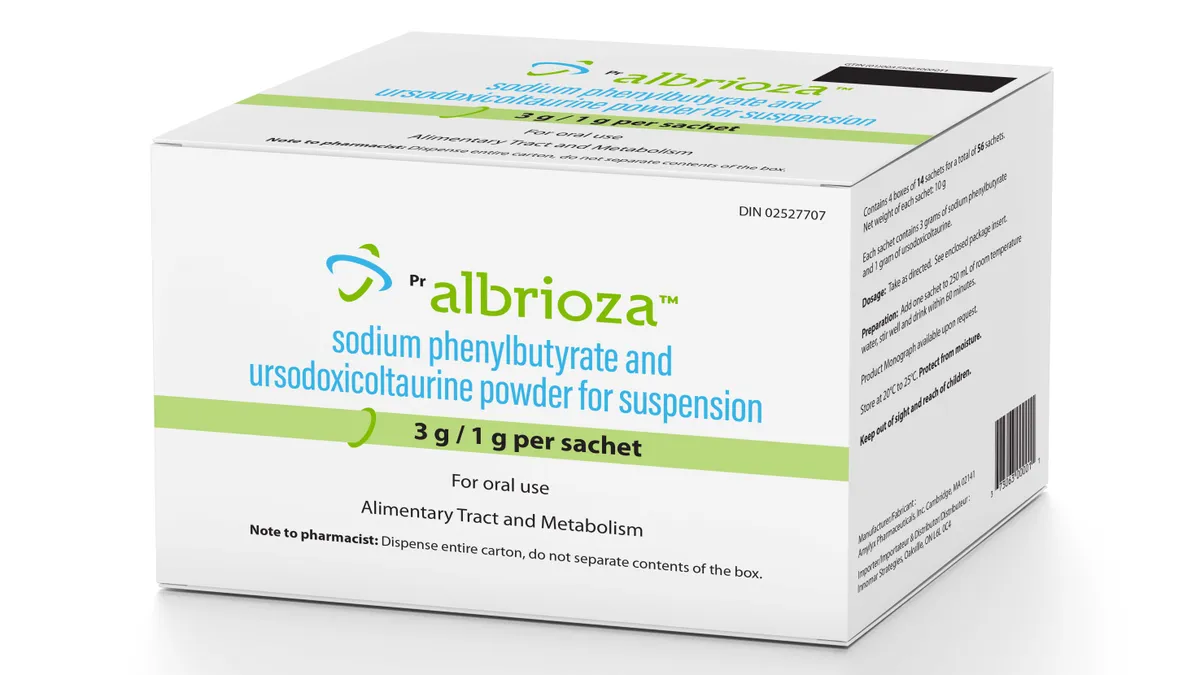
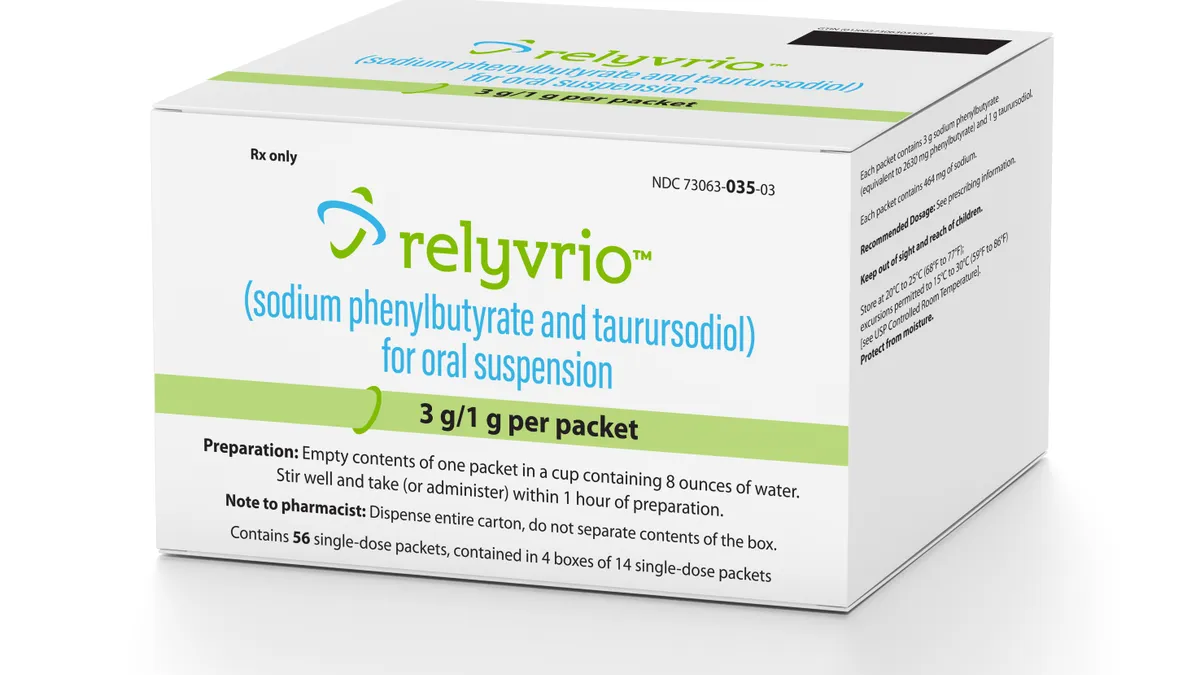
- The medicine is already approved in the U.S. and Canada, where Amylyx sells it under the brand names Relyvrio and Albrioza, respectively. The main evidence backing Albrioza comes from a clinical trial that enrolled about 140 patients with ALS, or amyotrophic lateral sclerosis, and found those given the drug rather than a placebo declined a small, but significant amount less, while also living a median five months longer.
- Yet, those results haven’t convinced a key regulatory committee that the medicine should be approved in Europe. This group, known in short as the CHMP, voted against Albrioza in June. Amylyx subsequently asked for a re-examination of its marketing application, but disclosed Friday that the previous negative opinion was affirmed. The company is running another, larger study of Albrioza, which it hopes will provide the evidence needed to take another shot at European approval.
Dive Insight:
The CHMP’s conclusion might not come as much of a surprise to Amylyx and its investors. In May, the company said it had been informed the committee was “trending toward a negative opinion.” And when that opinion came the following month, it was critical of Albrioza.
Regulators found the survival outcomes seen in that main trial to be “not reliable, given the way the data were collected and analyzed.” More broadly, they concluded a “positive balance of benefits and risks ... could not be established.”
Amylyx disagreed with the verdict, and requested a re-examination of its application — a process that takes about four months and appoints a different “rapporteur” and “co-rapporteur” from the initial evaluation.
With the negative opinion now affirmed, Amylyx faces a longer road to getting its drug approved in Europe, which, by the company’s estimates, is home to more than 30,000 ALS patients.
“ALS has no geographical boundaries, and we are working with urgency toward providing timely, broad, and sustainable access to [Albrioza] for eligible people living with ALS who may benefit,” said Amylyx’s co-founders and co-CEOs, Joshua Cohen and Justin Klee, in a statement.
“We share the frustration felt by the European ALS community, who has no time to wait for new, safe, and effective treatment options,” added Stéphanie Hoffmann-Gendebien, the company’s head of Europe, Middle East and Africa markets. “We remain committed to exploring all potential paths forward in support of the company’s goal of getting [Albrioza] to people living with ALS in the EU as quickly as possible.”
That larger study of Albrioza remains ongoing, with the aim of collecting more data on the drug’s safety and effectiveness in treating ALS. It has enrolled more than 660 participants, and results are expected in the middle of next year. If they’re supportive, Amylyx says it will use them to again seek approval in the European Union.
In the U.S., Amylyx has priced a year’s supply of its drug at $158,000, which is similar to other marketed ALS treatments but also far higher than what one prominent watchdog of drug costs believes is reasonable. That price, combined with strong demand among patients, have led to sales that exceeded Wall Street expectations. Over the first six months of this year, Amylyx recorded $170 million in net product revenue.
However, the challenges securing European approval appear to have affected some investors. Amylyx’s share price dipped around 7% when the CHMP delivered the initial negative opinion in June. And on Friday, it was down 3% by late morning, hovering just above $17.