An experimental drug developed by Eli Lilly significantly slowed mental and physical decline in a large clinical trial of people with early Alzheimer’s disease, the company said Wednesday in a result that brings closer a new treatment for the most common cause of dementia.
Lilly plans to submit an application for Food and Drug Administration approval of the drug, called donanemab, before the end of June, and said it would work with regulators to “achieve the fastest path” to clearance in the U.S. and elsewhere.
The trial, begun in 2020 and called Trailblazer-Alz 2, has been closely watched as a definitive test of donanemab ever since earlier study results suggested the drug could delay Alzheimer’s progression. Lilly enrolled more than 1,700 people with early forms of the disease, who either received the drug or placebo and were followed for 18 months.
Results released by Lilly Wednesday show that, on the trial’s main measure, people who received donanemab declined 35% slower than those given placebo. The measure, known in shorthand as iADRS, was designed by Lilly to assess cognition, as well as the ability to perform daily tasks like managing finances or self-care.
Importantly, the iADRS results nearly mirrored a 36% slower decline for donanemab-treated patients on another measure, called CDR-SB, that has been widely used in previous Alzheimer’s disease trials and is more familiar to physicians.
Nearly half of trial participants on donanemab experienced no decline on the CDR-SB measure after one year, compared to 29% of people in the placebo group.
Full results will be presented at the Alzheimer’s Association International Conference in July, and will be published in a medical journal, the company said.
Lilly designed its study carefully, having learned from past failures with other Alzheimer’s drugs. It only enrolled people who had plaques of a sticky protein called amyloid, and also evidence of buildup of another protein known as tau. Participants in the trial received donanemab until imaging scans showed a certain amount of amyloid had been cleared.
According to the company, 52% of people on the drug were able to complete treatment by one year, rising to 72% by 18 months.
Lilly focused its main analysis on the roughly 1,200 study participants who had low or intermediate levels of tau in their brains, which is associated with the earlier stages of disease. But it also looked at the overall study population, including people who had high tau concentrations. Across this larger group, treatment slowed decline on iADRS by 22% compared to placebo, and by 29% on CDR-SB.
While the results are a rare Alzheimer’s study success, they also show donanemab comes with side effects that may make regulators and physicians cautious. About one-quarter of patients on the drug had brain swelling associated with a type of imaging abnormality called ARIA, and 31% showed signs of micro-bleeds alongside those abnormalities.
Serious cases of ARIA occurred in 1.6% of study patients and three patients died, including two whose deaths were attributed to the side effect.
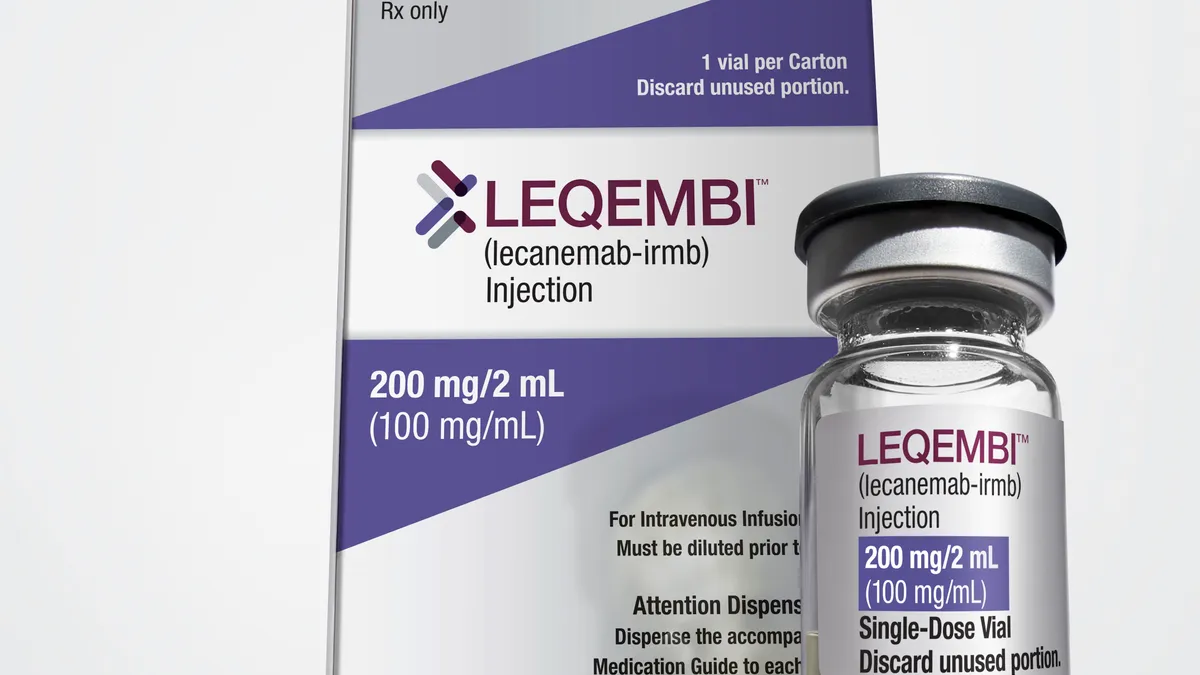
Lilly’s results are likely to be compared to those for Leqembi, a similar type of Alzheimer’s drug developed by Eisai and Biogen that won an accelerated FDA approval in January. More recent results from a Phase 3 trial showed Leqembi slowed Alzheimer’s decline by 27% compared to a placebo on the CDR-SB scale.
However, the companies’ trial did not separate participants by tau levels, making it more difficult to conclude whether donanemab is more effective, according to Wall Street analysts who cover Biogen and Lilly.
Leqembi does appear to be associated with lower rates of ARIA. In that recent study, researchers reported brain swelling in 13% of patients.
For Lilly, donanemab’s success is a culmination of around two decades of work to test whether removing amyloid from the brains of Alzheimer’s patients might slow or even stop the course of the disease. Academic researchers and other, earlier drug developers have been pursuing this “amyloid hypothesis” even longer.
Multiple large trials of a previous Lilly drug called solanezumab failed over the past decade, as did dozens of trials of other drugs targeting amyloid, deepening doubts about the protein’s role in Alzheimer’s progression. In response, drugmakers like Lilly, Eisai and Biogen developed more potent drugs and tested them earlier, hoping to forestall decline before more serious symptoms begin.
The first fruits of those efforts was a Biogen drug called aducanumab that showed promise, only to deliver contradictory results in late-stage trials. The FDA approved it anyway in a controversial 2021 decision, but it has been little used since due to doubts over whether it works.
Leqembi has a stronger case. Still, its use has been constrained by a restrictive reimbursement policy by Medicare, which covers most of the people eligible for the drug in the U.S.
That policy could soon change, however. After Eisai and Biogen released the Leqembi data, the Centers for Medicare and Medicaid Services indicated it could revise its rules following “any new evidence that becomes available.”
Shares in Lilly rose by as much as 5% Wednesday morning on the news, briefly pushing the company’s market value past $400 billion.
Editor’s note: This story has been updated with additional details throughout.