Dyno Therapeutics, a Harvard University spinout working to improve gene therapy, is expanding with $100 million in new funding from tech investor Andreessen Horowitz and several other venture firms.
Since officially launching a year ago, Dyno has struck deals with Novartis, Roche and Sarepta Therapeutics to aid their efforts to develop more efficient delivery of gene therapy. But even after doubling in size to 50 employees, the company hasn't been able to keep up with demand for its services, CEO Eric Kelsic said in an interview.
"Even though it's in the very beginning, we just had so many folks who want to partner with us," said Kelsic. "This is really to meet the demand that we've seen for partnering and to enable us to both expand our [existing] partnerships, as well as to work with new partners."
Over the next two years, Dyno hopes to triple its workforce, adding employees on both the scientific side to work with partners on research as well as on the business side to develop and support those collaborations. The Series A round announced Thursday will help fund that expansion.
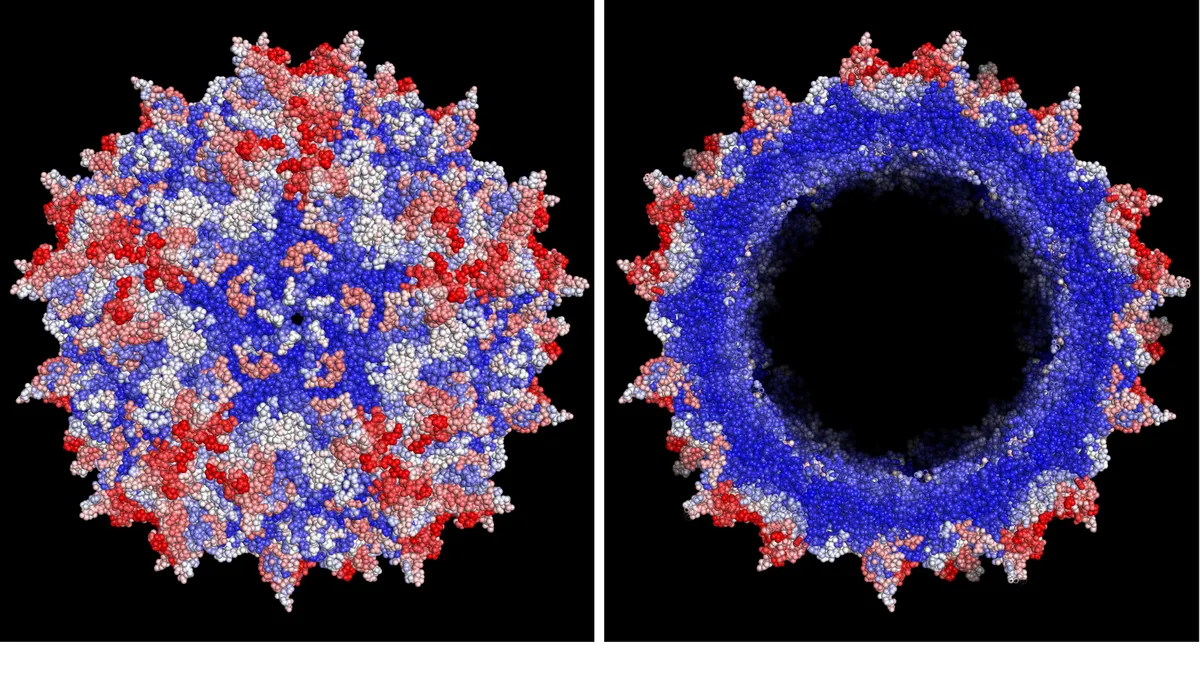
Dyno emerged last May to take forward technology built by the Harvard Wyss Institute for Biologically Inspired Engineering. It aims to solve some of the limitations of adeno-associated viruses, one of the main delivery vehicles for shuttling genetic material into human cells.
People can have pre-existing immunity to the protein shell, known as a capsid, that surrounds those viruses, for example. Infusion can also trigger immune responses. Other limitations include the efficiency by which genetic material is delivered into cells, the capacity capsids have to carry that material and the types of tissue the capsids can reach.
To get around those challenges, Dyno is leaning on machine learning, combined with data from in vivo research, to optimize AAVs better suited for carrying therapeutic DNA.
Engineering better AAVs is a goal that a number of new startups share, including Affinia Therapeutics and Taysha Gene Therapies. In Dyno's case, however, the company plans to design capsids for its partners rather than developing treatments itself.
All three companies are now flush with new cash, underscoring the growing investor interest. Taysha went public with a $181 million initial public offering in September 2020 and Affinia on Monday announced a $110 million venture funding round.
Manufacturing and quality control is a particular challenge for gene therapy developers and, in the past year, the Food and Drug Administration has appeared to apply closer scrutiny. Both Sarepta and competitor Pfizer, for example, have encountered delays on key research programs for Duchenne muscular dystrophy treatments due to questions on tests they use to measure their product's potency. Others have been set back by FDA requests for more information on their production processes.
Custom-designed capsids can help, Kelsic said. "Scale up is a challenge. In some cases, you need to scale up the dose in order to treat the disease and in other cases it's to scale up to treat other patients who need to benefit."
"Our platform directly helps with that," he added. "By making the delivery more efficient, that means you can actually treat the disease more effectively with a lower dose. With the efficiency being higher you can make more doses per batch and therefore treat more patients."
Deals with Novartis and Sarepta, which were announced when the company launched publicly last May, target muscle and eye diseases, respectively. Together, the two collaborations could earn Dyno up to $2 billion in partnership revenue, depending on how well the projects that emerge from them advance.
Roche, which bought the gene therapy pioneer Spark Therapeutics in 2019, came along in October with a partnership aiming at neurological and liver diseases. That deal could be worth up to $1.8 billion in milestone payments.
And if Dyno's expansion plans pay off, more announcements could be coming still.