When Misty Lovelace was a baby, her eyes were drawn to the light.
She could not focus on faces, only sources of light. Her grandmother Cynthia Lovelace, who would become her main caretaker, suspected vision problems.
By age three, Misty was diagnosed as legally blind. School systems struggled with how to handle her. She was intelligent and intuitive, but people would treat her as if she had a learning disability.
As she got older, Misty started carrying a lamp with her at school. She would put her lunch under it to see what she was about to eat. She learned Braille and used a cane to navigate. When she visited the doctor for checkups, her prognosis seemed to get worse.
"[The doctor] would take her little face and he'd put his hands on her face and say, 'Misty, I'm so sorry, there's nothing more we can do for you, honey. You're going to wake up in the dark one day,'" Lovelace recalled.
"It'd be like looking through a tunnel. And all of a sudden that tunnel goes out."
Misty has Leber congenital amaurosis, or LCA, a genetic disorder that often manifests at a young age, causing vision loss. In Misty's case, and for approximately 1,000 to 2,000 other people in the U.S., the disease is caused by mutations in a gene called RPE65.
What Misty didn't know as her vision got darker was that a scientist and doctor duo at the Children's Hospital of Philadelphia had already spent years working on a gene therapy for her disease.
The gene therapy, which would eventually become known as Luxturna, was not an overnight success. Decades of research and setbacks preceded the landmark U.S. approval of Luxturna four years ago, the first the Food and Drug Administration had ever granted to a gene therapy for an inherited disease. While Luxturna is not a cure for blindness, treatment has brought sustained improvements in sight, particularly in lower light, for several patients who spoke with BioPharma Dive. As a result, they've needed less help in educational and social environments, and have more independence.
Their experience with Luxturna is proof of gene therapy's potential as well as its limitations. As the first gene therapy of its kind, Luxturna also holds lessons for a field that's grown dramatically since its December 2017 approval.
A gene therapy first
Lovelace said she never stopped trying to find a way for Misty to regain her sight. The possibility gave her hope as she watched her granddaughter adjust to a life that, for her, was almost in total darkness.
A call from Jean Bennett was a lifeline.
Bennett and her husband, Albert Maguire, met at Harvard Medical School in the early 1980s. The two began researching gene therapy together, attempting to treat blindness in mice. Soon they were testing their approach on Briard dogs with the same defective RPE65 gene that causes LCA in humans.
By 2007, their gene therapy was ready to be tested in people — a high-stakes proposition for a field that had largely been shut down nearly a decade before. After 18-year-old Jesse Gelsinger died during a 1999 gene therapy study, many questioned whether such research was safe. The success Bennett and Maguire had with Luxturna was a large part of gene therapy's journey back to the forefront of biomedical research, aided by improvements in how such treatments are designed and delivered.
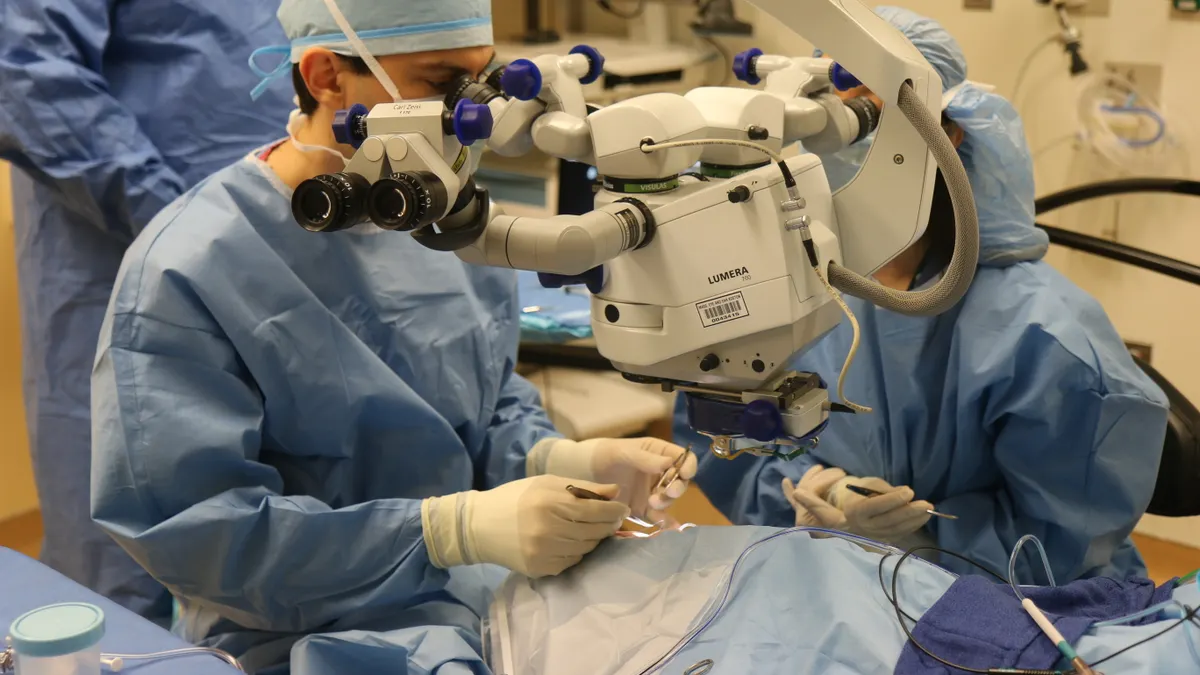
Testing began at the Children's Hospital of Philadelphia, where Misty was recruited as a study participant. At age 12, she took her first flight out of Kentucky and received the gene therapy in both eyes, starting with the one with worse vision.
"We didn't know if I was going to get worse, stay the same or get better," she said. "But in my mind, I was going to be completely blind by 18, so what's knocking a couple years off?"
The improvements were almost immediate, however. Lovelace recalls her granddaughter commenting on her wrinkles as soon as the eye patches from the procedure were removed. Misty could make out the fine hairs on the manes of horses, her favorite animal and hobby. Rainbows and stars, though, she found underwhelming.
More than eight years later, Misty says she's grateful she "took the leap," attributing to Luxturna her independence and ability to pursue a career as a horse trainer.
Results from early participants like Misty led to the formation of Spark Therapeutics and a larger clinical trial in Pennsylvania and at the University of Iowa that gave the biotech company the evidence needed to approach the FDA.
On Oct. 12, 2017, a panel of scientists and FDA advisers unanimously endorsed the gene therapy, with Misty one of several individuals who shared their stories. The FDA followed with an approval on Dec. 18, a gene therapy milestone.
"For many of us, this is exactly the type of disease that we hoped that gene therapy would someday treat," Wilson Bryan, director of an FDA office tasked with reviewing Luxturna, said at the time. The next year, Luxturna was also approved in Europe.
It's unclear how many people have received Luxturna since. A Spark spokesperson told BioPharma Dive the company does not disclose that information. In 2019, the company told the Philadelphia Business Journal it had shipped 75 vials of the gene therapy in its first year post-approval. (One vial is used per eye.)
Spark is now owned by the Swiss pharmaceutical company Roche, which does not disclose sales of Luxturna. In February, however, Roche reduced the accounting value of Luxturna, citing "reduced sales expectations."
'This is not a cure'
Luxturna consists of one hundred and fifty billion copies of the corrected RPE65 gene encoded into modified viruses, which are delivered into the eye via about 0.3 milliliters of liquid. Those few drops are injected underneath the retina and, over the course of a week, the viral particles shuttle the functional gene into the patient's eye cells. Once inside, the gene instructs the cells to produce a protein that's otherwise missing, helping restore visual function.
"This is not a cure," said Jason Comander, a physician at Massachusetts Eye and Ear in Boston who has administered Luxturna. "It will not make your vision normal," he added, "and there's a small chance that it could hurt your vision." Comander consults with other drugmakers and in 2019 received a nominal amount from Spark.
Luxturna also benefits each patient differently. Comander said the vast majority gain some night vision, while others report improvements in central or side vision. Some see more substantial improvements — one of his patients was able to see in up to one thousand times dimmer light than in pre-surgery exams. Many have been able to walk without canes and read without using Braille after surgery.
Their vision isn't perfect, however. Some recipients, Misty included, are still considered legally blind and unable to drive. How long the benefit of gene therapy treatment will last is still unclear, though a recent study co-authored by Maguire and Bennett indicated "improvements were maintained up to 3 to 4 years" after Luxturna.
Comander, who was in his residency while Luxturna was tested, said seeing Maguire administer the therapy affirmed his decision to go into the practice. Now, Comander has done close to a dozen surgeries; his youngest patient was 4 years old at the time of treatment and his oldest was in their 30s. While younger patients saw greater improvements, each patient's eyes functioned better in lower light following treatment.
For Comander, Luxturna was an inspiration, one that he said has helped fuel greater interest in gene therapy. "Many careers have been dedicated to expanding on the success of Luxturna, and it's made a huge difference in the field," he said.
Since Luxturna's clearance, Novartis won FDA approval in May 2019 for a spinal muscular atrophy treatment known as Zolgensma, making it the second gene therapy for an inherited disease available in the U.S. A handful of other gene therapies are in late-stage testing and, behind them, are an expanding pipeline of experimental medicines for a constellation of genetic conditions. In 2020 alone, the FDA received more than 230 applications from cell and gene therapy developers to begin clinical trials, the head of the agency's biologic drugs division said earlier this year.
"It's like he's a new kid every day"
Gordon "Creed" Pettit was one of the kids who couldn't get into clinical trials for Luxturna. His mother, Sarah St. Pierre-Pettit, brought him from Florida to the University of Iowa a number of times. But he couldn't get through the tests needed to qualify him for treatment.
From there, it was a waiting game until Luxturna's approval. Soon after the FDA's decision, Pierre-Pettit brought Creed to Audina Berrocal at the Bascom Palmer Eye Institute in Miami.
Creed was Berrocal's first Luxturna patient. As a pediatric retina specialist, Berrocal said Spark sought her out in the fall of 2017. To date, she's performed a dozen surgeries, all of which have yielded positive results.
"Of all the things I've done in my career, this has been the most amazing and the most rewarding in the sense that we are changing the genetics, the DNA of a person, and we're allowing them to do things that before they couldn't do," Berrocal said. Berrocal consults with other drugmakers and has contributed to published research on Luxturna. In 2018 and 2019, she received nominal payments from Spark.
But treatment, even when positive, can come with adjustments, too. In Creed's case, he was overwhelmed by the sudden change, at first telling his mother he wished he had his old eyes back.
With time, however, Creed has started challenging himself more. "I think most of the gains were at the beginning," Pierre-Pettit said. "Whatever Luxturna did is done. But now that he finally feels confident with himself, he's putting Luxturna to the test now."
For Creed, that means being more social and inquisitive about the world around him. Now 12 years old, he hasn't mentioned wanting his old eyes back for years.
"It's still almost like a new kid every day, like a new baby that sees something new," his mother said.
A sky-high price tag
From a young age, Luke Ward told his mother, Stephanie Joachim, about his dream of playing soccer. But the sport — as well as many other daily tasks — seemed out of reach.
His vision problems were apparent from birth. While his twin sister could track people with her eyes, Luke stared only at sources of light. When he started walking, he needed to put his hands out to stop himself from running into walls.
Genetic testing revealed Luke had LCA. His doctor said he'd be legally blind by kindergarten. Around the same time, Joachim read an article about Luxturna, but was too late to get Luke enrolled in clinical testing. By the time the FDA approved the therapy, the family had already decided that Luke was getting Luxturna.
But Joachim was anxious after learning Luxturna's price tag of $425,000 per eye. "I was just flabbergasted and I was like, 'You know what, it's fine. We have the best health insurance,'" she said.
To the family's disappointment, and as other Luxturna patients have experienced, insurance denied the request and cited the therapy's then "newness" as a reason.
At some point in the process, however, Luke's file crossed the desk of an anonymous person who was "so moved from Luke's story and from Luke's pictures, he volunteered to pay for Luke's surgery," Joachim said.
Luxturna's cost was criticized when the therapy was approved and has remained an issue within the patient community since. Shortly after the FDA gave its OK, Spark announced a program with health insurer Harvard Pilgrim and affiliates of Express Scripts, through which the company agreed to pay rebates if the drug doesn't help patients meet certain thresholds.
In a statement to BioPharma Dive, Spark said it offers a "range of patient services and payment models to help navigate and support access" to Luxturna, but did not respond to questions on the number of times rebates have been paid.
"Parents shouldn't be paying for this out of pocket," Berrocal, who was also Luke's surgeon, said.
Berrocal told Luke he's the "poster child for Luxturna," Joachim said. He can play sports with his twin sister, including soccer and tee-ball. He started kindergarten this year and has no issues seeing the whiteboard. He still has visual impairments, though, including his peripheral vision. His mother says they keep their shoes tucked out of the way in the house to prevent Luke from tripping.
"This is what we have, and it's working"
Four years after its approval, Luxturna continues to be sought out by patients. Joachim says she's received messages from people in Spain, South Africa and the U.K. inquiring about Luke and his progress.
And as Luxturna keeps working, other drugmakers hope to replicate its success. The eye, in particular, is the focus of many gene therapy developers, as it's easy to access and targeting it doesn't carry as many safety risks as other organs. Novartis, which sells Luxturna in Europe, AbbVie, Biogen and Johnson & Johnson are all exploring gene therapies for the eye.
Research into gene editing is advancing as well. In September, Editas Medicine shared preliminary results from the first trial testing a CRISPR gene editing treatment that does its work inside the body. Treatment appeared safe, although the efficacy results were mixed, with several patients experiencing little improvement in vision. The treatment uses CRISPR editing to restore the function of eye cells in people with another form of LCA known as type 10.
Berrocal believes Luxturna represents the beginning of what genetic medicine can offer to patients with many inherited diseases, not only those of the eye.
"20 years from now, we could look back and say, 'Oh my god, that was so rudimentary. Look how much you have advanced,'" she said. "But we have to start somewhere, right? And in 2021, this is what we have, and it's working."