Marketing: Page 16
-
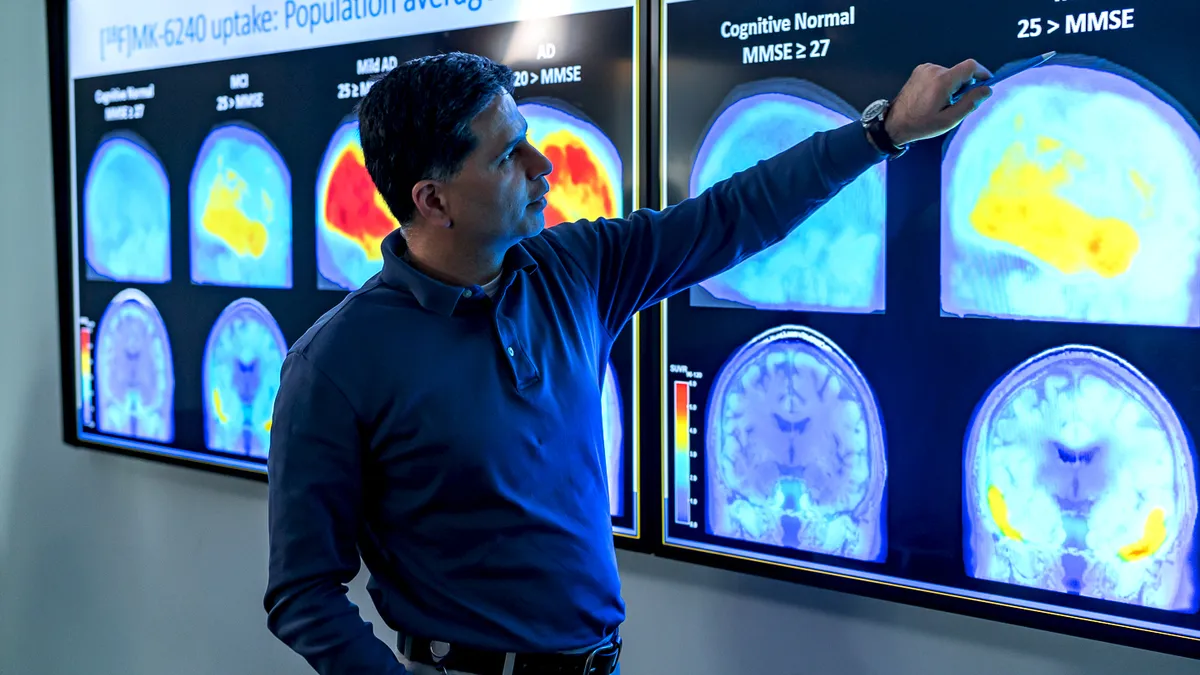
After controversial approval, doctors are still debating how to use Biogen's Alzheimer's drug
Nearly two months since Aduhelm became available, many physicians have yet to use the first treatment approved in the U.S. to slow Alzheimer's disease.
By Jacob Bell • Updated Aug. 3, 2021 -
Sponsored by Health Union
Social health: The future of healthcare marketing
Approximately 79% of patients look to connect through social media to find answers to their health questions.
July 26, 2021 -
 Explore the Trendline➔
Explore the Trendline➔
 Brian Tucker / BioPharma Dive/BioPharma Dive
Brian Tucker / BioPharma Dive/BioPharma Dive Trendline
TrendlineCommercialization
New drugs for obesity are becoming blockbusters, while Trump administration pressure is reshaping pharma marketing strategies ahead of looming patent cliffs.
By BioPharma Dive staff -
Major health centers, insurers push back against Aduhelm
The Cleveland Clinic and Mount Sinai won't yet administer the controversial Alzheimer's drug, while UnitedHealth's CEO said the insurer would need more time to iron out coverage.
By Jacob Bell • July 15, 2021 -
Q&A
Biohaven CEO on migraine drug's fast launch, TikTok ads and battling big pharma
Sales of Nurtec ODT during the second quarter were double what Wall Street expected. CEO Vlad Coric explained to BioPharma Dive how the biotech did it, despite being far smaller than its chief rival.
By Jacob Bell • July 14, 2021 -
Medicare to weigh nationwide coverage rules for Biogen Alzheimer's drug
CMS will consider whether to set a single policy for Aduhelm, a process that could shape how millions of Americans access the controversial treatment.
By Ned Pagliarulo • Updated July 13, 2021 -
Alzheimer's Association, insurers press Medicare for coverage rules on Aduhelm
Days after the FDA narrowed the target population for Biogen's drug, the patient advocacy group said it's urging Medicare to adopt a nationwide policy. Payers are seeking guidance, too.
By Jonathan Gardner • Updated July 12, 2021 -
Bluebird resumes marketing gene therapy in Europe
Zynteglo sales have been on hold since February, when a patient in a clinical trial of another, related Bluebird medicine developed leukemia.
By Jacob Bell • July 9, 2021 -
Biohaven beats expectations with fast sales of migraine drug
Second quarter sales of Nurtec ODT, which recently became the first migraine drug cleared for acute and preventive treatment, were nearly twice what analysts on Wall Street had predicted.
By Ned Pagliarulo • July 8, 2021 -
FDA revises labeling of Biogen's Alzheimer's drug to emphasize early treatment
Aduhelm's approval last month, already highly controversial, was made more so by the agency's decision to clear the drug for a broad group of patients. New prescribing information more closely matches clinical testing criteria.
By Ned Pagliarulo , Ben Fidler • July 8, 2021 -
WHO backs Roche, Sanofi drugs for COVID-19 after research ups and downs
Once long-shot experiments in the rush to repurpose drugs for COVID-19, both Actemra and Kevzara could become more widely used, renewing questions around cost and access.
By Ben Fidler • July 7, 2021 -
Nestlé joins Seres in deal to market microbiome drug
The health science unit at Nestlé may pay more than $500 million to secure rights in the U.S. and Canada for SER-109, a spore-filled capsule meant for patients with recurrent C. diff infections.
By Kristin Jensen • July 1, 2021 -
ICER stands by view on Biogen's Aduhelm, says sharp price cut needed
The influential nonprofit maintains that the clinical trial evidence supporting Aduhelm is insufficient to prove a health benefit. But if some efficacy is assumed, ICER argues Biogen's $56,000 price tag is far too high.
By Ned Pagliarulo • June 30, 2021 -
Sponsored by FedEx Office
How biopharma innovation may benefit from more inclusive print communications
Medical advancements are evolving at a rapid pace, and so is public consumption of information.
June 28, 2021 -
European regulators push 8 drugs toward approval, including controversial anemia pill
A key committee of the EMA gave a positive review of Astellas and Fibrogen's roxadustat, as well as Bristol Myers Squibb's cell therapy Abecma and UCB's psoriasis drug Bimzelx.
By Jacob Bell • June 25, 2021 -
As 5 Blue Cross Blue Shield plans join to tackle drug prices, some remain skeptical
Another healthcare venture promises to lower costs, this time by using patient data on drug efficacy through a handful of large insurers.
By Samantha Liss • June 23, 2021 -
Anthem joins insurer-backed generics effort CivicaScript
The subsidiary of hospital-owned nonprofit CivicaRx plans to make six to 10 common generic drugs that don't have enough competition to drive down cost.
By Rebecca Pifer Parduhn • June 16, 2021 -
Sponsored by 81qd
Clinical leadership: A foundational double attack strategy
We define the most influential clinicians as Clinical Leaders, those who measurably impact the clinical behavior of other clinicians in their "clinical networks."
June 14, 2021 -
Deep Dive // New Alzheimer's drugs
A first-of-its-kind Alzheimer's drug raises heavy questions around who will and won't get it
Biogen priced its newly approved medicine Aduhelm at an average cost of $56,000 a year, adding affordability to other barriers patients may face.
By Jacob Bell • June 8, 2021 -
Sponsored by IQVIA
Moving pharma marketing from multichannel to omnichannel
IQVIA experts weigh in on the importance of an omnichannel approach.
June 7, 2021 -
FDA restricts use of Intercept drug due to liver injury risk
An investigation linked the drug to severe injuries in about two dozen patients, leading the agency to add a new contraindication to the label.
By Kristin Jensen • May 27, 2021 -
ICER, vocal critic of drug company pricing, turns scrutiny to insurers
The watchdog group plans to examine how cost sharing can hurt access to care, but it won't assess some of the most controversial insurer practices.
By Jonathan Gardner • May 25, 2021 -
Pfizer, BioNTech to send EU up to 1.8B coronavirus vaccine doses
The deal, which the companies and the bloc have been negotiating for weeks, will significantly expand on the current contract for 600 million shots.
By Kristin Jensen • May 20, 2021 -
Humira patents take center stage as House panel targets AbbVie pricing
CEO Richard Gonzalez defended the company's practices, arguing its profits help spur innovation: "The products that are on the market today pay for the products of the future," he said.
By Jonathan Gardner • May 18, 2021 -
Sponsored by FedEx Office
6 tips to deliver information that matters using print communications
As remote work continues, print is as important as ever for biopharmaceutical companies when it comes to reaching a varied audience that may include patients, doctors' offices, researchers, and internal staff.
May 17, 2021 -
Sponsored by IQVIA
Top customer engagement trends in pharma
The future of provider and patient engagement in healthcare.
May 17, 2021